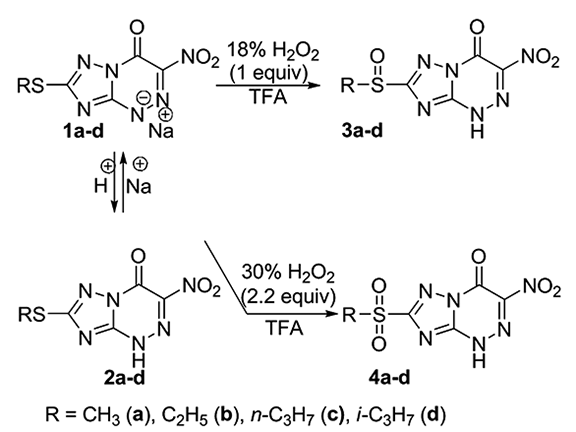
The redox transformations and nucleophilic replacements as possible metabolic reactions of the drug “Triazaverin”. The chemical modeling of the metabolic processes
Abstract
Keywords
Full Text:
PdfReferences
Rusinov VL, Ulomskii ЕN, Chupakhin ОN, Charushin VN. Azolo[5,1-c]-1,2,4-triazines as a new class of antiviral compounds. Russ Chem Bull, Int Ed. 2008;57(5):985–1014. doi:10.1007/s11172–008–0130–8
Karpenko I, Deev S, Kiselev O, Charushin V, Rusinov V, Ulomsky E, Deeva E, Yanvarev D, Ivanov A, Smirnova O, Kochetkov S, Chupakhin O, Kukhanova M. Antiviral properties, metabolism, and pharmacokinetics of a novel azolo-1,2,4-triazine-derived inhibitor of influenza A and B virus replication. Antimicrob Agents Chemother. 2010;54(5):2017–22. doi:10.1128/AAC.01186–09
Denicola A, Radi R. Peroxynitrite and drug-dependent toxicity. Toxicology. 2005;208(2):273–88. doi:10.1016/j.tox.2004.11.023
Liu J., Dang Q., Wei Z., Zhang H., Bai X. Parallel solution-phase synthesis of a 2,6,8,9-tetrasubstituted purine library via a sulfur intermediate. J Comb Chem. 2005;7(4):627–36. doi:10.1021/cc049819p
Rusinov VL, Ulomskii ЕN, Chupakhin ОN, Petrov АYu, Sharonov ЕА. Nitroazines. 9. Characteristic features of nucleophilic substitution of the nitro group in dihydroazolo[5,1-c] [1,2,4]triazines. Chem Heterocycl Compd. 1989;25(2):209–13. doi:10.1007/BF00479921
DOI: https://doi.org/10.15826/chimtech.2015.2.3.025
Copyright (c) 2015 N. R. Medvedeva, I. S. Sapozhnikova, V. L. Rusinov, E. N. Ulomsky

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice






