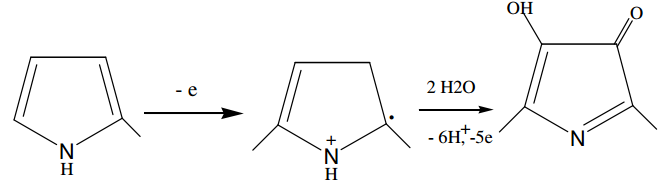
The mathematical description of the electrosynthesis of composites of oxy-hydroxycompounds cobalt with polypyrrole overooxidazed
Abstract
Keywords
Full Text:
PDFReferences
Sadki S, Scottland Ph, Brodie N, Saboraud G. The mechanisms of pyrrole electropolymerization. Chem Soc Rev. 2000;29(5):283-9. doi:10.1039/A807124A
Roncali J. Conjugated poly(thiophenes): Synthesis, functionalization and applications. Chem Rev. 1992;92:4; 711-738. doi:10.1021/cr00012a009
Bodnaryuk-Lupshak N, Aksimentyeva OI. Voprosy khimii i khimicheskoy tekhnologii. 50;1998:1. Russian.
Yamamoto T. Molecular assembly and properties of polythiophenes. NPG Asia Materials. 2010;2:54-60. doi:10.1038/asiamat.2010.37
Ojani R, Raoof JB, Ahmady A, Hosseini SR. Highly improved methanol oxidation onto carbon paste electrode modified by nickel particles dispersed into poly (2,5-dimethylaniline) film. Caspian Journal of Chemistry [Internet]. 2013[cited 2016];2(1):45-57. Available from: http://caschemistry.journals.umz.ac.ir/article_510_13d7ba627fde246025b83b64cbda0a4f.pdf
de Andrade VM. Confecção de biossensores através da imobilização de biocomponentes por eletropolimerização de pirrol [master's thesis]. Porto Alegre (Brazil): Universidade Federal do Rio Grande do Sul; 2006. 112 p. Portuguese. Available from: http://www.lume.ufrgs.br/bitstream/handle/10183/10393/000597791.pdf
Tosar Rovira JP. Estudio de la inmovilización de oligonucleótidos a eléctrodos modificados de oro:polipirrol, y detección electroquímica de secuencias complementarias [master's thesis]. Montevideo (Uruguay): Universidad de la República; 2008. Spanish.
Ramanavicius A, Ramanaviciene A,Malinauskas A. Electrochemical sensors based on conducting polymer-polypyrrole. Electrochim Acta. 2006. 51;27:6025-37. doi:10.1016/j.electacta. 2005.11.052
Singh RP. Prospects of Organic Conducting Polymer Modified Electrodes: Enzymosensors. Int J Electrochem. 2012;2012:502707. doi:10.1155/2012/502707
Zane D, Appetecchi GB, Bianchini C, Passerini S, Curulli A. An impedimetric glucose biosensor based on overoxidized polypyrrole thin film. Electroanalitica. 2011;23(5):1134-41. doi:10.1002/elen.201000576
McQuade T, Pullen A, Swager TM. Conjugated polymer based chemical sensors. Chem Rev. 2000;100(7):2537-74. doi:10.1021/cr9801014
Ojani R, Raoof JB, Rahemi V. A simple and efficient electrochemical sensor for electrocatalytic reduction of nitrite based on poly94-aminoacetanilide) film using carbon paste electrode. J Chin Chem Soc. 2011;58(2):247-54. doi:10.1002/jccs.201190084
Sargentelli V, Ferreira AP. Magnetic nanoparticles: The cobalt. Ecletica Química. 2010;35(4):153-63. doi:10.1590/S0100-46702010000400020
Jamison TF, Shambayati S, Crowe WE, Schreiber SL. Tandem use of cobalt-mediated reactions to synthesize (+)-epoxydictymene, a diterpene containing a trans-fused 5-5ring system. J Am Chem Soc. 1997;119(19):4353-63. doi:10.1021/ja970022u
Stadnik O. [dissertation]. Kiev; 2011. Russian.
Ivanova N, Stadnik OA, Boldyrev E. Oscillations in the Electrodeposition of Cobalt Oxide Compounds. In: I4 Oscillations and Pattern Formation in Electrochemistry. 218th ECS Meeting Abstracts; 2010 Oct 2-15; Las Vegas (NV). ECS: 2010. No. MA2010-02 2240.
Ansari R. Polypyrrole Conducting Electroactive Polymers: Synthesis and Stability Studies. E-Journal of Chemistry. 2006;3(4):186-206. doi:10.1155/2006/860413
Ba-Shammakh MS, Rahman SU, Abul-Hamayel MA, Kahraman R. Thermal effects on the process of electropolymerization of pyrrole on mild steel. In: AC1 Organic and Bioorganic Electrochemistry General Session. 203rd ECS Meeting Abstracts; 2003 April 27 - May 2; Paris, France. No. 2468. Available from: http://www.electrochem.org/dl/ma/203/pdfs/2468.pdf
Liu AS, Oliveira MAS. Electrodeposition of polypyrrole films on aluminum from tartrate aqueous solution. J Braz Chem Soc. 2007;18(1):143-52. doi:10.1590/S0103-50532007000100016
Sazou D. The dynamical behavior of the electrochemical polymerization of indole on Fe in acetonitrile-water mixtures. Synth Met. 2002;130(1)45-54. doi:10.1016/S0379-6779(02)00110-8
Das I, Goel N, Agrawal NR, Gupta SK. Growth patterns of dendrimers and electric potential oscillations during electropolymerization of pyrrole using mono- and mixed surfactants. J Phys Chem B. 2010;114(40):12888-96. doi:10.1021/jp105183q
Bazzaoui M, Bazzaoui EA, Martins L, Martins JI. Electropolymerization of pyrrole on zinc-lead-silver alloys electrodes in acidic and neutral organic media. Synth Met. 2002;130(1):73-83. doi:10.1016/S0379-6779(02)00101-7
Das I, Goel N, Gupta SK, Agrawal NR. Electropolymerization of pyrrole: Dendrimers, nano-sized patterns and oscillations in potential in presence of aromatic and aliphatic surfactants. J Electroanal Chem. 2012;670(1):1-10. doi:10.1016/j.jelechem.2012.01.023
Aoki K, Mukoyama I, Chen J. Compettion between polymerization and dissolution of poly(3-methylthiophene) films. Russ J Electrochem. 2004;40(3):280-5. doi:10.1023/B:RUEL.0000019665.59805.4c
Das I, Agrawal NR, Ansari SA, Gupta SK. Pattern formation and oscillatory electropolymerization of thiophene. Ind J Chem A. 2008;47(12):1798-803. Available from: http://nopr.niscair.res.in/bitstream/123456789/2565/1/IJCA%2047A%2812%29%201798-1803.pdf
Ba-Shammakh MS. Electropolymerization of pyrrole on mild steel for corrosion protection [dissertation]. Dharan (Saudi Arabia): King Fahd University of Petroleum and Minerals; 2002.
Pagitsas M, Dimitra S. The improved Franck-FitzHung model for the electrodissolution of iron in sulphuric acid solutions: linear stability and bifurcation analysis. Derivation of the kinetic equations for the forced franck-FitzHung models. Electrochim Acta. 1991;36(8):1301-8. doi:10.1016/0013-4686(91)80009-w
Pearlstein AJ, Johnson JA. Global and conditional stability of the steady and periodic solutions of the Franck-FitzHugh model of electrodissolution of Fe in H2SO4. J Electrochem Soc. 1989;136(5):1290-9. doi:10.1149/1.2096909
Ogüz Güler M et al. In: 7th Turkish Nanoscience and Nanotechnology Conference Proceedings. 2011. p. 24.
Roncali J. Conjugated poly(thiophenes): Synthesis, functionalization and applications. Chem Rev. 1992;92(4):711-38. doi:10.1021/cr00012a009
Lemos Castagno KR. [dissertation]. Porto Alegre (Brazil): Universidade Federal do Rio Grande do Sul; 2006.
Mostany J, Scharifker BR. Impedance spectroscopy of undoped, doped and overoxidazrd polypyrrole films. Synth Met. 1997;87(3):179-85. doi:10.1016/S0379-6779(97)80105-1
Tkach VV, Nechyporuk VV, Yagodynets´ PI, Rusnak I. [The CP-based electrochemical biosensors with autocatalytic stage in their function and the mathematical description of their work]. Orbital: Electron J Chem [Internet]. 2012[cited 2016];4(2):136-45. Spanish. Available from: http://www.orbital.ufms.br/index.php/Chemistry/article/view/280/pdf
Tkach V, Nechyporuk V, Slipenyuk O. Eclética Química. 2012;37:72. Spanish.
Tkach VV, de Rocha AM. Naukoviy visnik Chernivets'kogo universitetu. 2013;658:132. Ukrainian.
Tkach V, Nechyporuk V, Yagodynets P. Electropolymerization of heterocyclic compounds. Mathematical models. Ciência & Tecnologia dos Materiais. 2012;24(1-2):54-8.
Tkach V, Nechyporuk V, Yagodynets P. Proc Quím. 2014;8:49. Spanish.
Tkach V, Nechyporuk V, Yagodynets P, Hryhoryak M. Cien Tecn Mat. 2012;24:50.
Tkach V, Nechyporuk V, Yagodynets P. Tecn Met Mat Min. 2013;10:249.
Tkach V, Nechyporuk V, Yagodynets P. Descripción matemática de la síntesis electroquímica de polímeros conductores en la presencia de surfactantes. Avances en Química [Internet]. 2013[cited 2016];8(1):9-15. Spanish. Available from: http://erevistas.saber.ula.ve/index.php/avancesenquimica/article/download/6357/6168
Tkach V, Nechyporuk V, Yagodynets´ P. [The general mathematical model for the oscillatory behavior of the electrochemical systems with electropolymerization of heterocyclic compounds in organic solvents in passivation conditions]. Avances en Química [Internet]. 2013[cited 2016];8(2):97-104. Spanish. Available from: http://erevistas.saber.ula.ve/index.php/avancesenquimica/article/download/6348/6162
Tkach V, Nechyporuk V, Yagodynets´ P. Estudio matemático de las condiciones de estabilidad en la polimerización electroquímica de compuestos heterocíclicos en presencia de oxidantes. Afinidad [Internet]. 2013[cited 2016];70(561):70-4. Catalan. Available from: http://www.raco.cat/index.php/afinidad/article/download/268543/356131
Tkach VV, Nechyporuk VV, Yagodynets PI, de Oliveira SC, de Rocha AM. Matematicheskoye opisaniye elektropolimerizatsii elektrokhimicheski modifitsirovannykh monomerov v gal'vanostaticheskom rezhime [The Mathematical Description for the electropolymerization of electrochemical modified monomers in Galvanostatic Mode]. Zhurnal sibirskogo federal'nogo universiteta [Journal of Siberian Federal University]. 2014;7:403-11. Russian. Available from: http://elib.sfu-kras.ru/bitstream/handle/2311/13421/10_Tkach.pdf
Tkach V, Nechyporuk V, Yagodynets´ P. In: Anais do 10o Encontro Brasileiro sobre Adsorção. 2014 Apr 27-30; Guarujá (SP). No. 122-1. Spanish.
Tkach V, Nechyporuk V, Yagodynets P. The mathematical description for the electropolymerization of furan, pyrrole and thiophene derivatives in alkaline media. Mediterr J Chem [Internet]. 2015[cited 2016];3(6):1122-8. Available from: http://www.medjchem.com/index.php/medjchem/article/download/185/pdf_6
Tkach VV, Nechyporuk VV, Yagodynets PI. The mathematical stability study of the work of isomerization sensors, based on conduction polymers. Anal Bioanal Electrochem [Internet]. 2014[cited 2016];6(3):273-83. Available from: http://www.abechem.com/No.%203-2014/2014,6_3_273-283.pdf
Tkach V, Swamy BK, Ojani R, Blanes M, Yagodynets P. El Mecanismo de la Oxidación de Omeprazol Sobre el Electrodo de Carbono Vitroso, Modificado por Polializarina, y Su Descripción Matemática. Orbital: Electron J Chem [Internet]. 2015;7(1):1-4. Spanish. doi:10.17807/orbital.v7i1.599
Tkach V, Ojani R, Nechyporuk V, Yagodynets P. Revista Colombiana de Ciencias Químico. 2014;43:35.
Tkach VV, Ojani R, Nechyporuk V, Yagodynets´ PI. A Descrição Matemática do Desempenho Eletroanalítico de Nanopartículas de Óxido de Cobre(II) na Eletrodetecção de Hidrazina. Orbital: Electron J Chem [Internet]. 2014[cited 2016];6(3):142-6. Portuguese. Available from: http://www.orbital.ufms.br/index.php/Chemistry/article/view/583/pdf
DOI: https://doi.org/10.15826/chimtech.2016.3.1.003
Copyright (c) 2016 V. V. Tkach, S. C. de Oliveira, J. Maia, B. G. Soares, R. Ojani, P. I. Yagodynets

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice






