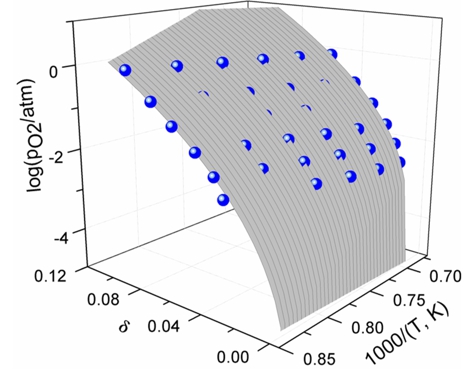
Oxygen non-stoichiometry and defect structure of LaMn1-zCuzO3+δ
Abstract
Keywords
Full Text:
PDFReferences
Jonker GH, Van Santen JH. Ferromagnetic compounds of manganese with perovskite structure. Physics. 1950;16:337–49. doi:10.1016/0031-8914(50)90033-4
Menzler NH, Tietz F, Uhlenbruck S, Buchkremer HP, Stöver D. Materials and manufacturing technologies for solid oxide fuel cells. J Mater Sci. 2010;45:3109-35. doi:10.1007/s10853-010-4279-9
Van Santen JH, Jonker GH. Electrical conductivity of ferromagnetic compounds of manganese with perovskite structure. Physics. 1950;16:599–600. doi:10.1016/0031-8914(50)90104-2
Royer S, Duprez D. Catalytic Oxidation of Carbon Monoxide over Transition Metal Oxides. ChemCatChem. 2011;3(1):24–65. doi:10.1002/cctc.201000378
Chan KS, Ma J, Jaenicke S, Chuah GK, Lee JY. Catalytic carbon monoxide oxidation over strontium, cerium and copper-substituted lanthanum manganates and cobaltates. Appl Catal, A. 1994;107:201–7. doi:10.1016/0926-860X(94)85156-5
Petrov AN, Zuev AYu, Tikchonova IL, Voronin VI. Crystal and defect structure of the mixed oxides LaMn1-zCuzO3±y (0≤z≤0.4). Solid State Ionics. 2000;129(1-4):179-88. doi:10.1016/S0167-2738(99)00324-0
Jonker GH. Magnetic and Semiconducting Properties of Perovskites Containing Manganese and Cobalt. J Appl Phys. 1966;37:1424-30. doi:10.1063/1.1708498
Zuev AYu, Tsvetkov DS. Oxygen nonstoichiometry, defect structure and defect-induced expansion of undoped perovskite LaMnO3±δ. Solid State Ionics. 2010;81(11-12):557–63. doi:10.1016/j.ssi.2010.02.024
Porta P, De Rossi S, Faticanti M, Minelli G, Pettiti I, Lisi L, Turco M. Perovskite-Type Oxides I. Structural, Magnetic, and Morphological Properties of LaMn1-xCuxO3-d and LaCo1-xCuxO3-d Solid Solutions with Large Surface Area. J Solid State Chem. 1999;146(2):291-304. doi:10.1006/jssc.1999.8326
Tabata K, Hirano Y, Suzuki E. XPS studies on the oxygen species of LaMn1-xCuxO3+λ. Appl Catal, A. 1998;170:245–54. doi:10.1016/S0926-860X(98)00062-3
DOI: https://doi.org/10.15826/chimtech.2015.2.4.031
Copyright (c) 2015 A. Yu. Zuev, D. S. Tsvetkov

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice






