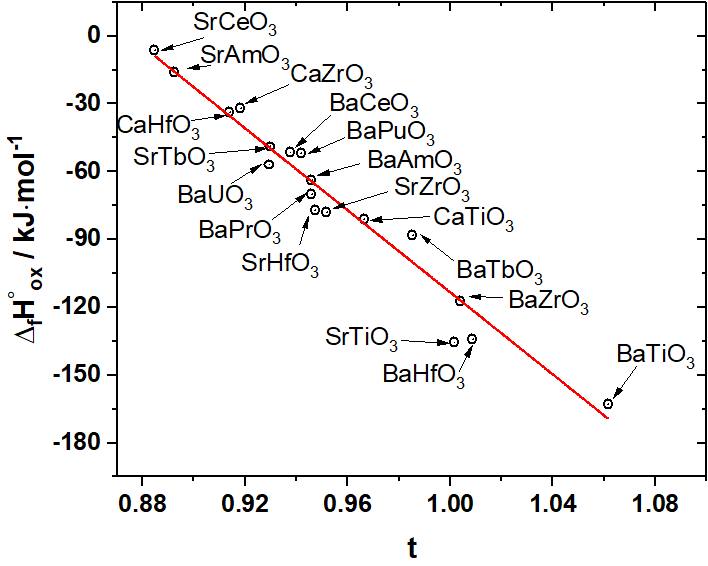
Thermodynamics of formation of solid solutions between BaZrO3 and BaPrO3
Abstract
Keywords
Full Text:
PDFReferences
Kreuer KD. Proton-Conducting Oxides. Annu Rev Mater Res. 2003;33(1):333–59. doi:10.1146/annurev.matsci.33.022802.091825
Norby T. Proton Conductivity in Perovskite Oxides. Boston, MA: Springer US; 2009. 217 p. (Ishihara T, editor. Perovskite Oxide for Solid Oxide Fuel Cells). doi:10.1007/978-0-387-77708-5_11
Sažinas R, Einarsrud M-A, Grande T. Toughening of Y-doped BaZrO3 proton conducting electrolytes by hydration. J Mater Chem A. 2017;5(12):5846–57. doi:10.1039/C6TA11022C
Iguchi F, Tsurui T, Sata N, Nagao Y, Yugami H. The relationship between chemical composition distributions and specific grain boundary conductivity in Y-doped BaZrO3 proton conductors. Solid State Ion. 2009;6–8(180):563–8. doi:10.1016/j.ssi.2008.12.006
Babilo P, Uda T, Haile SM. Processing of yttrium-doped barium zirconate for high proton conductivity. J Mater Res. 2007;22(5):1322–30. doi:10.1557/jmr.2007.0163
Ryu KH, Haile SM. Chemical stability and proton conductivity of doped BaCeO3–BaZrO3 solid solutions. Solid State Ion. 1999;125(1):355–67. doi:10.1016/S0167-2738(99)00196-4
Duval SBC, Holtappels P, Vogt UF, Pomjakushina E, Conder K, Stimming U, Graule T. Electrical conductivity of the proton conductor BaZr0.9Y0.1O3−δ obtained by high temperature annealing. Solid State Ion. 2007;178(25):1437–41. doi:10.1016/j.ssi.2007.08.006
Kjølseth C, Fjeld H, Prytz Ø, Dahl P, Estournès C, Haugsrud R, Norby T. Space–charge theory applied to the grain boundary impedance of proton conducting BaZr0.9Y0.1O3−δ. Solid State Ion. 2010;181. doi:10.1016/j.ssi.2010.01.014
Magrasó A, Frontera C, Gunnæs AE, Tarancón A, Marrero-López D, Norby T, Haugsrud R. Structure, chemical stability and mixed proton–electron conductivity in BaZr0.9−xPrxGd0.1O3−δ. J Power Sources. 2011;196(22):9141–7. doi:10.1016/j.jpowsour.2011.06.076
Fabbri E, Markus I, Bi L, Pergolesi D, Traversa E. Tailoring mixed proton-electronic conductivity of BaZrO3 by Y and Pr co-doping for cathode application in protonic SOFCs. Solid State Ion. 2011;202(1):30–5. doi:10.1016/j.ssi.2011.08.019
Fabbri E, Bi L, Tanaka H, Pergolesi D, Traversa E. Chemically Stable Pr and Y Co-Doped Barium Zirconate Electrolytes with High Proton Conductivity for Intermediate-Temperature Solid Oxide Fuel Cells. Adv Funct Mater. 2011;21(1):158–66. doi:10.1002/adfm.201001540
Tsvetkov D, Sednev-Lugovets A, Malyshkin D, Sereda V, Zuev A, Ivanov I. Crystal structure and high-temperature thermodynamic properties of Pr-doped barium zirconates, BaZr1-xPrxO3 (x = 0.1, 0.5). J Phys Chem Solids. Forthcoming 2020.
Huntelaar ME, Booij AS, Cordfunke EHP. The standard molar enthalpies of formation of BaZrO3(s) and SrZrO3(s). J Chem Thermodyn. 1994;26(10):1095–101. doi:10.1006/jcht.1994.1127
Gonçalves MD, Maram PS, Muccillo R, Navrotsky A. Enthalpy of formation and thermodynamic insights into yttrium doped BaZrO3. J Mater Chem A. 2014;2(42):17840–7. doi:10.1039/C4TA03487B
Katsura T, Kitayama K, Sugihara T, Kimizuka N. Thermochemical Properties of Lanthanoid-Iron-Perovskite at High Temperatures. Bull Chem Soc Jpn. 1975;48(6):1809–11. doi:10.1246/bcsj.48.1809
Katsura T, Sekine T, Kitayama K, Sugihara T, Kimizuka N. Thermodynamic properties of Fe-lathanoid-O compounds at high temperatures. J Solid State Chem. 1978;23(1):43–57. doi:10.1016/0022-4596(78)90052-X
Petrov AN, Kropanev AY, Zhukovskij BM. Thermodynamic properties of rare earth cobaltites, RCoO3. Zhurnal Fizicheskoj Khimii. 1984;58(1):50–3.
Navrotsky A. Energetics of Phase Transition in AX, ABO3 and AB2O4 Compounds. New York: Academic Press; 1981. 71 p. (O’Keefe M., Navrotsky A., Editors. Structure and Bonding in Crystals).
Shannon RD. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr Sect A. 1976;32(5):751–67. doi:10.1107/S0567739476001551
Ushakov SV, Cheng J, Navrotsky A, Wu JR, Haile SM. Formation Enthalpies of Tetravalent Lanthanide Perovskites by High Temperature Oxide Melt Solution Calorimetry. MRS Online Proceedings Library Archive. 2002;718. doi:10.1557/PROC-718-D7.17
Morss LR, Mensi N. Enthalpy of Formation of Barium Lanthanide(IV) Oxides: BaCeO3, BaPrO3, and BaTbO3. Boston, MA: Springer; 1982. 279 p. (G.J. McCarthy, H.B. Silber, J.J. Rhyne, Editors. The Rare Earths in Modern Science and Technology). doi:10.1007/978-1-4613-3406-4_56
NIST Standard Reference Database Number 69 [Internet]. Washington: National Institute of Standards and Technology; 2018 [modified October 2018; cited 2020 April 26]. Available from: https://doi.org/10.18434/T4D303
Todd SS, Lorenson RE. Heat Capacities at Low Temperatures and Entropies at 298.16°K. of Metatitanates of Barium and Strontium. J Am Chem Soc. 1952;74(8):2043–5. doi:10.1021/ja01128a054
Navrotsky A. Thermochemistry of crystalline and amorphous phases related to radioactive waste. Netherlands: Kluwer Academic Publishers; 1998. 267 p. (P.A. Sterne, A. Gonis, A.A. Borovoi, Editors. Actinides and the environment)
SpringerMaterials [Internet]. New York; 2020 [modified 2020 April 28; cited 2020 April 28]. Available from: https://materials.springer.com/
Termicheskie konstanty veshestv [Internet]. Moscow: Moscow State University; 2020 [modified 2020 April 28; cited 2020 April 28]. Available from: http://www.chem.msu.ru/cgi-bin/tkv.pl
Goudiakas J, Haire RG, Fuger J. Thermodynamics of lanthanide and actinide perovskite-type oxides IV. Molar enthalpies of formation of MM′O3 (M = Ba or Sr, M′ = Ce, Tb, or Am) compounds. J Chem Thermodyn. 1990;22(6):577–87. doi:10.1016/0021-9614(90)90150-O
Antunes I, Amador U, Alves A, Correia MR, Ritter C, Frade JR, Pérez-Coll D, Mather GC, Fagg DP. Structure and Electrical-Transport Relations in Ba(Zr,Pr)O3−δ Perovskites. Inorg Chem. 2017;56(15):9120–31. doi:10.1021/acs.inorgchem.7b01128
Jacobson AJ, Tofield BC, Fender BEF. The structures of BaCeO3, BaPrO3 and BaTbO3 by neutron diffraction: lattice parameter relations and ionic radii in O-perovskites. Acta Cryst B. 1972;28(3):956–61. doi:10.1107/S0567740872003462
Glasser L, Jenkins HDB. Predictive thermodynamics for condensed phases. Chem Soc Rev. 2005;34(10):866–74. doi:10.1039/B501741F
Kurosaki K, Konings RJM, Wastin F, Yamanaka S. The low-temperature heat capacity and entropy of SrZrO3 and BaZrO3. J Alloys Compd. 2006;424(1):1–3. doi:10.1016/j.jallcom.2005.09.096
Ahrens M, Maier J. Thermodynamic properties of BaCeO3 and BaZrO3 at low temperatures. Thermochim Acta. 2006;443(2):189–96. doi:10.1016/j.tca.2006.01.020
Cordfunke EHP, van der Laan RR, van Miltenburg JC. Thermophysical and thermochemical properties of BaO and SrO from 5 to 1000 K. J Phys Chem of Solids. 1994;55(1):77–84. doi:10.1016/0022-3697(94)90186-4
Konings RJM, Beneš O, Kovács A, Manara D, Sedmidubský D, Gorokhov L, et al. The Thermodynamic Properties of the f-Elements and their Compounds. Part 2. The Lanthanide and Actinide Oxides. J Phys Chem Ref Data. 2014;43(1):013101. doi:10.1063/1.4825256
DOI: https://doi.org/10.15826/chimtech.2020.7.2.01
Copyright (c) 2020 Dmitry Tsvetkov, Vladimir Sereda, Dmitry Malyshkin, Anton Sednev-Lugovets, Andrey Zuev, Ivan Ivanov

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice







