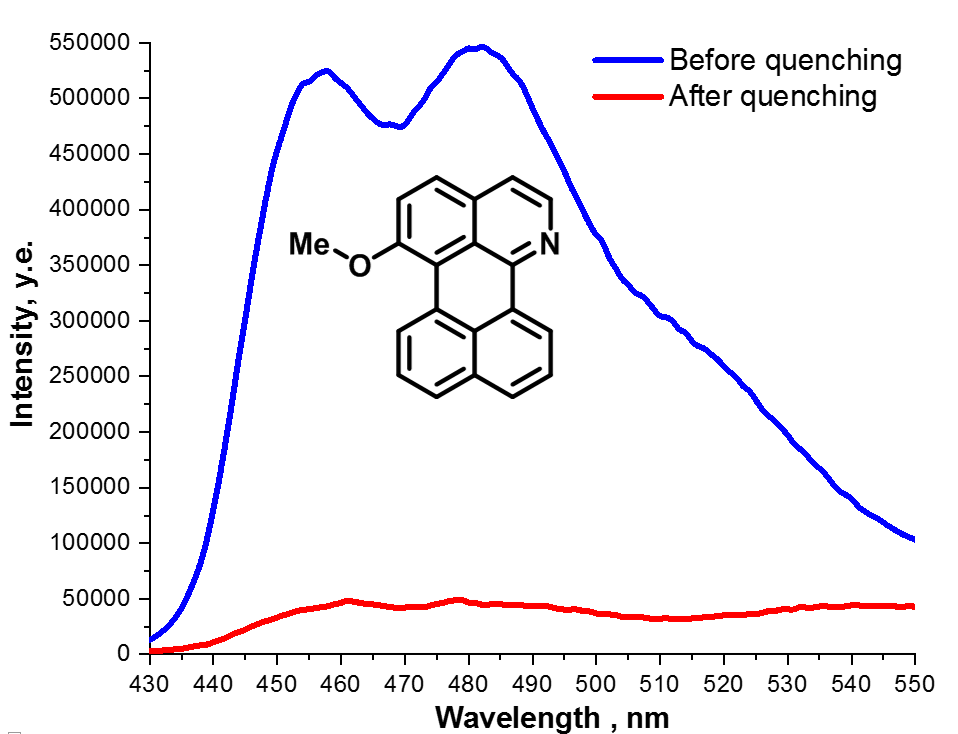
Benzo[de]naphtho[1,8-gh]quinolines: synthesis, photophysical studies and nitro explosives detection
Abstract
Keywords
Full Text:
PDFReferences
Zyryanov GV, Kopchuk DS, Kovalev IS, Nosova EV, Rusinov VL, Chupakhin ON. Chemosensors for detection of nitroaromatic compounds (explosives). Russ Chem Rev. 2014;83(9):783–819. doi:10.1070/RC2014v083n09ABEH004467
Lee JH, Rock JC, Schlautman MA, Carraway ER. Characteristics of key intermediates generated in uncatalyzed bis(2,4-dinitrophenyl) oxalate (DNPO) chemiluminescence reactions. J Chem Soc, Perkin Trans. 2002;2:1653–7. doi:10.1039/b206367k
Pandey S, Fletcher KA, Powell JR, McHale MER, Kauppila A-SM, Acree Jr WE, Fetzer JC, Dai W, Harvey RG. Spectrochim. Acta Part A. 1997;53:165–72.
Zhang P-F, Zeng J-C, Zhuang F-D, Zhao K-X, Sun Z-H, Yao Z-F, Lu Y, Wang X-Y, Wang J-Y, Pei J. Parent B2N2-perylenes with different BN orientations. Angew Chem Int Ed. 2021;60:23313–9. doi:10.1002/anie.202108519
Cui X, Zhao J, Yang P, Sun J. Zinc(II) tetraphenyltetrabenzoporphyrin complex as triplet photosensitizer for triplet–triplet annihilation upconversion. Chem Commun. 2013;49:10221–3. doi:10.1039/c3cc45843a
Hirono A, Sakai H, Hasobe T. Synthesis and electrochemical and photophysical properties of azaterrylene derivatives. Chem Asian J. 2019;14:1754–62. doi:10.1002/asia.201801410
Harwell JR, Glackin JME, Davis NJLK, Gillanders RN, Credgington D, Turnbull GA, Samuel IDW. Sensing of explosive vapor by hybrid perovskites: effect of dimensionality. APL Mater. 2020;8:071106. doi:10.1063/5.0011229
Neese F. The ORCA program system. WIREs Comput Mol Sci. 2012;2:73–78. doi:10.1002/wcms.81
Tóth J, Dancsó A, Blaskó G, Tőke L, Groundwater PW, Nyerges M. 1,7-Electrocyclization reactions of stabilized α,β:γ,δ-unsaturated azomethine ylides. Tetrahedron. 2006;62:5725–35. doi:10.1016/j.tet.2006.03.088
Min L, Yang W, Weng Y, Zheng W, Wang X, Hu Y. A method for Bischler–Napieralski-type synthesis of 3,4-dihydroisoquinolines. Org Lett. 2019;21(8):2574–7. doi:10.1021/acs.orglett.9b00534
Gryko DT, Piechowska J, Gałȩzowski M. Strongly emitting fluorophores based on 1-azaperylene scaffold. J Org Chem. 2010;75:1297–30. doi:10.1021/jo902443s
Porrès L, Holland A, Palsson L-O, Monkman AP, Kemp C, Beeby A. Absolute measurements of photoluminescence quantum yields of solutions using an integrating sphere. J Fluoresc. 2006;16:267–73. doi: 10.1007/s10895-005-0054-8
Krishnan R, Binkley JS, Seeger R, Pople JA. Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys. 1980;72:650–4. doi:10.1063/1.438955
McLean AD, Chandler J. GS. Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z=11–18. Chem. Phys. 1980;72:5639–48. doi:10.1063/1.438980
Clark T, Chandrasekhar J, Schleyer PvR. Efficient diffuse function-augmented basis sets for anion calculations. III. The 3-21+G basis set for first-row elements, Li–F. J Comp Chem. 1983;4:294–301. doi:10.1002/jcc.540040303
Frisch MJ, Pople JA, Binkley JS. Self-consistent molecular orbital methods 25. Supplementary functions for Gaussian basis sets. J Chem Phys. 1984;80:3265–9. doi:10.1063/1.447079
Mohamad M, Ahmed R, Shaari A, Goumri‑Said S. Structure‑dependent optoelectronic properties of perylene, di‑indenoperylene (DIP) isolated molecule and DIP molecular crystal. Chem Cent J. 2017;11:125. doi:10.1186/s13065-017-0352-7
Shrivastava A, Gupta V. Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chronicles Young Sci. 2011;2:21. doi:10.4103/2229-5186.79345
DOI: https://doi.org/10.15826/chimtech.2021.8.4.15
Copyright (c) 2021 Igor L. Nikonov, Igor A. Khalymbadzha, Leila К. Sadieva, Maria I. Savchuk, Ekaterina S. Starnovskaya, Dmitry S. Kopchuk, Igor S. Коvalev, Grigory А. Kim, Oleg N. Chupakhin

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice







