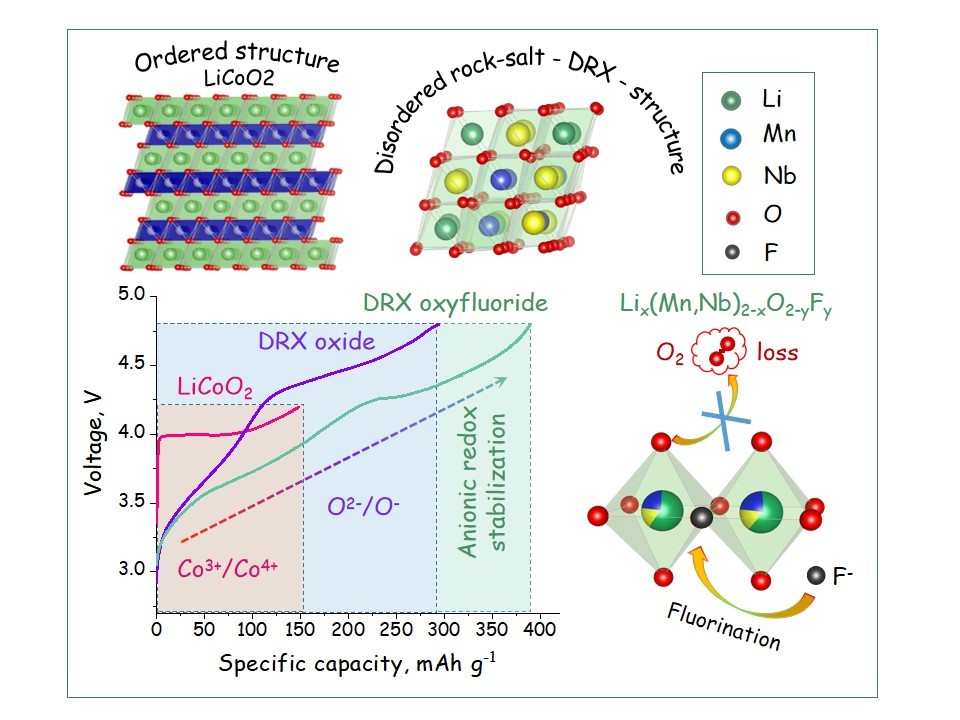
Effect of cooling rate on the structure and electrochemical properties of Mn-based oxyfluorides with cation-disordered rock-salt structure
Abstract
Keywords
Full Text:
PDFReferences
Lee J, Urban A, Li X, Su D, Hautier G, Ceder G. Unlocking the potential of cation-disordered oxides for rechargeable lithium batteries. Sci. 2014;343:519–522. doi:10.1126/science.1246432
Yabuuchi N. Material design concept of lithium-excess electrode materials with rocksalt-related structures for rechargeable non-aqueous batteries. Chem Rec. 2019;19:690–707. doi:10.1002/tcr.201800089
Lun Z, Ouyang B, Kwon DH, Ha Y, Foley EE, Huang TY, Cai Z, Kim H, Balasubramanian M, Sun Y, Huang J, Tian Y, Kim H, McCloskey BD, Yang W, Clément RJ, Ji H, Ceder G. Cation-disordered rock-salt-type high-entropy cathodes for Li-ion batteries. Nat Mater. 2021;20:214–221. doi:10.1038/s41563-020-00816-0
Lee J, Kitchaev DA, Kwon DH. Lee CW, Papp JK, Liu YS, Lun Z, Clément RJ, Shi T, McCloskey BD, Guo J, Balasubramanian M, Ceder G. Reversible Mn2+/Mn4+ double redox in lithium-excess cathode materials. Nat. 2018;556:185–190. doi:10.1038/s41586-018-0015-4
Ji H, Urban A, Kitchaev DA, Kwon DH, Artrith N, Ophus C, Huang W, Cai Z, Shi T, Kim JC, Kim H, Ceder G. Hidden structural and chemical order controls lithium transport in cation-disordered oxides for rechargeable batteries. Nat Commun. 2019;10:592–600. doi:10.1038/s41467-019-08490-w
Ouyang B, Artrith N, Lun Z, Jadidi Z, Kitchaev DA, Ji H, Urban A, Ceder G. Effect of fluorination on lithium transport and short-range order in disordered-rocksalt-type lithium-ion battery cathodes. Adv Energy Mater. 2020;1903240. doi:10.1002/aenm.201903240
Jones MA, Reeves PJ, Seymour ID, Cliffe MJ, Dutton SE, Grey CP. Short-range ordering in a battery electrode, the ‘cation-disordered’ rocksalt Li1.25Nb0.25Mn0.5O2. Chem Commun. 2019;55:9027–9030. doi:10.1039/c9cc04250d
Kan WH, Deng B, Xu Y, Shukla AK, Bo T, Zhang S, Liu J, Pianetta P, Wang BT, Liu Y, Chen G. Understanding the effect of local short-range ordering on lithium diffusion in Li1.3Nb0.3Mn0.4O2 single-crystal cathode. Chem. 2018;4:2108–2123. doi:10.1016/j.chempr.2018.05.008
Semykina DO, Morkhova YeA, Kabanov AA, Mishchenko KV, Slobodyuk AB, Kirsanova MA, Podgornova OA, Shindrov AA, Okhotnikov KS, Kosova NV. Effect of transition metal cations on the local structure and lithium transport in disordered rock-salt oxides. Phys Chem Chem Phys. 2022;24:5823–5832. doi:10.1039/D1CP04993C
Geng F, Hu B, Li C, Zhao C, Lafon O, Trébosc J, Amoureux JP, Shen M, Hu B. Anionic redox reactions and structural degradation in a cation-disordered rock-salt Li1.2Ti0.4Mn0.4O2 Cathode material revealed by solid-state NMR and EPR. J Mater Chem A. 2020;8:16515–16526. doi:10.1039/d0ta03358h
Stoyanova R, Gorova M, Zhecheva E. EPR of Mn4+ in spinels Li1+xMn2−xO4 with 0≤x≤0.1. J Phys Chem Solids. 2000;61(4):609–614. doi:10.1016/S0022-3697(99)00244-9
Lun Z, Ouyang B, Kitchaev DA, Clément RJ, Papp JK, Balasubramanian M, Tian Y, Lei T, Shi T, McCloskey BD, Lee J, Ceder G. Improved cycling performance of Li-excess cation-disordered cathode materials upon fluorine substitution. Adv Energy Mater. 2019;9:1802959. doi:10.1002/aenm.201802959
Croguennec L, Bains J, Ménétrier M, Flambard A, Bekaert E, Jordy C, Biensan P, Delmas C. Synthesis of "Li1.1(Ni0.425Mn0.425Co0.15)0.9O1.8F0.2" materials by different routes: is there fluorine substitution for oxygen? J Electrochem Soc. 2009;156:A349. doi:10.1149/1.3080659
Lun Z, Ouyang B, Cai Z, Cle´ment RJ, Kwon DH, Huang J, Papp JK, Balasubramanian M, Tian Y, McCloskey BD, Ji H, Kim H, Kitchaev DA, Ceder G. Design principles for high-capacity Mn-based cation-disordered rocksalt cathodes. Chem. 2020;6:153–168. doi:10.1016/j.chempr.2019.10.001
Kitchaev DA, Lun Z, Richards WD, Ji H, Clément RJ, Balasubramanian M, Kwon DH, Dai K, Papp JK, Lei T, McCloskey BD, Yang W, Lee J, Ceder G. Design principles for high transition metal capacity in disordered rocksalt Li-ion cathodes. Energy Environ Sci. 2018;11:2159–2171. doi:10.1039/C8EE00816G
Ahn J, Chen D, Chen G. A fluorination method for improving cation-disordered rocksalt cathode performance. Adv Energy Mater. 2020;10:2001671. doi:10.1002/aenm.202001671
Crafton MJ, Yue Y, Huang TY, Tong W, McCloskey BD. Anion reactivity in cation-disordered rocksalt cathode materials: the influence of fluorine substitution. Adv Energy Mater. 2020;10:2001500. doi:10.1002/aenm.202001500
Renfrew SE, McCloskey BD. Residual lithium carbonate predominantly accounts for first cycle CO2 and CO outgassing of Li-stoichiometric and Li-rich layered transition metal oxides. J Am Chem Soc. 2017;139:17853−17860. doi:10.1021/jacs.7b08461
Freunberger SA, Chen Y, Peng, Z, Griffin JM, Hardwick LJ, Barde F, Novak P, Bruce PG. Reactions in the rechargeable Lithium-O2 battery with alkyl carbonate electrolytes. J Am Chem Soc. 2011;133:8040−8047. doi:10.1021/ja2021747
Tan H, Verbeeck J, Abakumov A, Tendeloo GV. Oxidation state and chemical shift investigation in transition metal oxides by EELS. Ultramicroscop. 2012;116:24–33. doi:10.1016/j.ultramic.2012.03.002
Lin L, Wang K, Azmi R, Wang J, Sarkar A, Botros M, Najib S, Cui Y, Stenzel D, Sukkurji PA, Wang Q, Hahn H, Schweidler S, Breitung B. Mechanochemical synthesis: route to novel rock-salt structured high-entropy oxides and oxyfluorides. J Mater Sci. 2020;55:16879–16889. doi:10.1007/s10853-020-05183-4
Stoyanova RK, Zhecheva EN, Gorova MY. EPR evidence on short-range Co/Mn order in LiCoMnO4 spinels. J Mater Chem. 2000;10:1377–1381. doi:10.1039/A909066E
Julien C, Gendron F, Ziolkiewicz S, Nazri GA. Electrical and ESR studies of lithium manganese oxide spinels. Mat Res Soc Symp Proc. 1998;548:187–195. doi:10.1557/PROC-548-187
Ivakin YD, Danchevskaya MN, Murav'eva GP. State of manganese ions in the structure of corundum synthesized in supercritical water fluid. Moscow Univ Chem Bull. 2011;66(3):151–158. doi:10.3103/S0027131411030060
Lee YJ, Wang F, Grey CP. 6Li and 7Li MAS NMR studies of lithium manganate cathode materials. J Am Chem Soc. 1998;120(48):12601–12613. doi:10.1021/ja9817794
Du LS, Schurko RW, Kim N, Grey CP. Solid-state 93Nb, 19F, and 113Cd nuclear magnetic resonance study of niobium oxyfluorides: characterization of local distortions and oxygen/fluorine ordering. J Phys Chem A. 2002;106:7876–7886. doi:10.1021/jp020457q
Zhou K, Zheng S, Ren F, Wu J, Liu H, Luo M, Liu X, Xiang Y, Zhang C, Yang W, He L, Yang Y. Fluorination effect for stabilizing cationic and anionic redox activities in cation-disordered cathode materials. Energy Storage Mater. 2020;32:234–243. doi:10.1016/j.ensm.2020.07.012.
Kan WH, Chen D, Papp JK, Shukla AK, Huq A, Brown CM, McCloskey BD, Chen G. Unravelling solid-state redox chemistry in Li1.3Nb0.3Mn0.4O2 single-crystal cathode material. Chem Mater. 2018;30:1655−1666. doi:10.1021/acs.chemmater.7b05036
Chen D, Wu J, Papp JK, McCloskey B, Chen G. Role of redox-inactive transition-metals in the behavior of cation-disordered rocksalt cathodes. Small. 2020;16:173–177. doi:10.1002/smll.202000656
Clement R, Lun Z, Ceder G. Cation-disordered rocksalt transition metal oxides and oxyfluorides for high energy lithium-ion cathodes. Energy Environ Sci. 2020;1:345–373. doi:10.1039/C9EE02803J
DOI: https://doi.org/10.15826/chimtech.2022.9.3.10
Copyright (c) 2022 Kseniya V. Mishchenko, Maria A. Kirsanova, Arseny B. Slobodyuk, Anna A. Krinitsyna, Nina V. Kosova

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice







