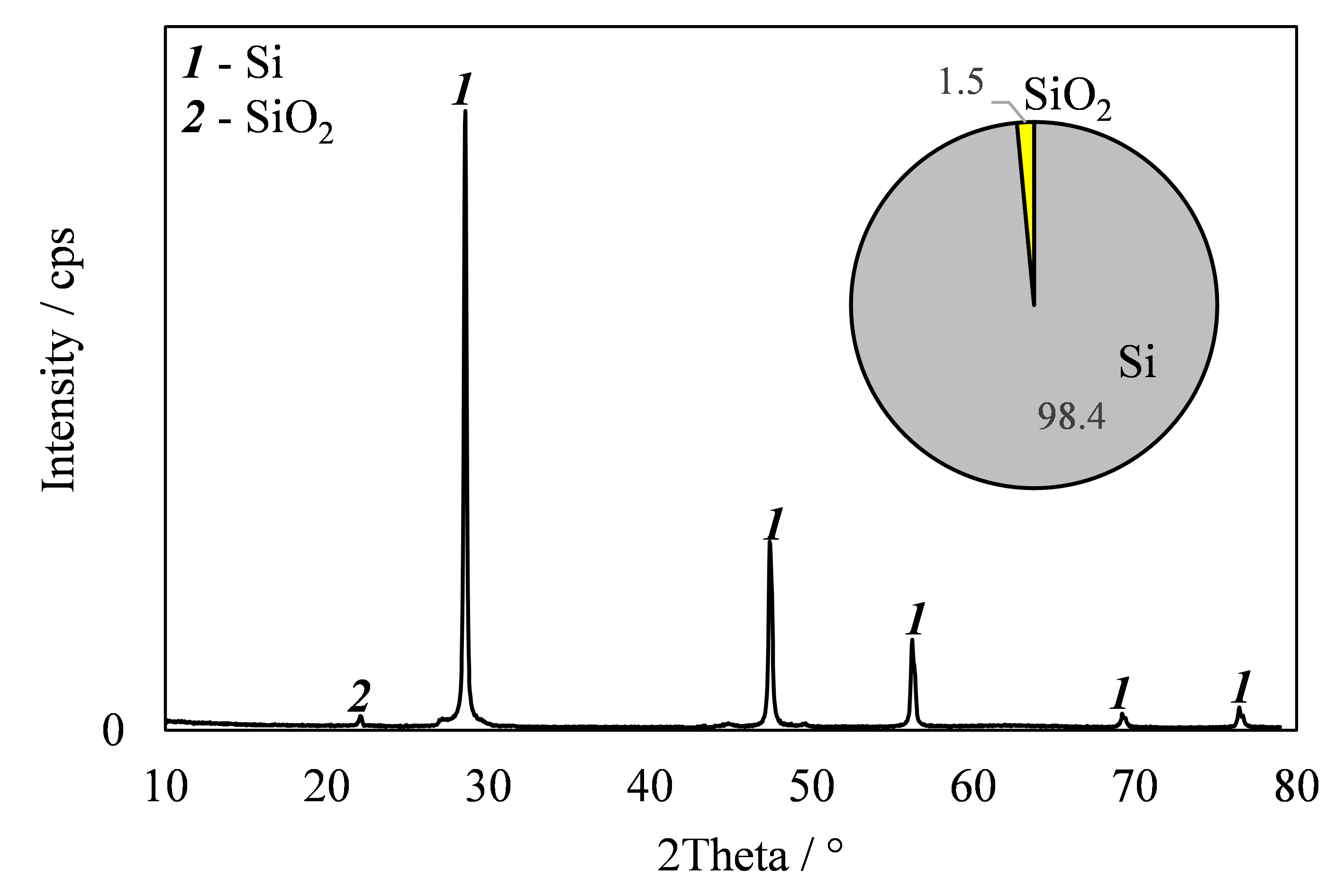
Electroreduction of silicon from the NaI–KI–K2SiF6 melt for lithium-ion power sources
Abstract
Keywords
Full Text:
PDFReferences
Cohen U. Some prospective applications of silicon electrodeposition from molten fluorides to solar cell fabrication. J Electron Mater. 1977;6:607–643. doi:10.1007/BF02660341
Gevel T, Zhuk S, Leonova N, Leonova A, Trofimov A, Suzdaltsev A, Zaikov Yu. Electrochemical synthesis of nano-sized silicon from KCl-K2SiF6 melts for powerful lithium-ion batteries. Appl Sci. 2021;11(22):10927. doi:10.3390/app112210927
Zh Yu, Fang Sh, Wang N, Shi B, Hu Y, Shi Zh, Shi D, Yang J. In-situ growth of silicon nanowires on graphite by molten salt electrolysis for high performance lithium-ion batteries. Mater Lett. 2020;273:127946. doi:10.1016/j.matlet.2020.127946
Islam MM, Said H, Hamzaoui AH, Fukata N, Akimoto K. Study of structural and optical properties of electrodeposited silicon films on graphite substrates. Nanomater. 2022;12(3):363. doi:10.3390/nano12030363
Fang D, Weimin Zh, Haiming Ya, Chengguo S, Geng X, Chen Y, Li L, Liu Z. Surface modification and functional structure space design to improve the cycle stability of silicon based materials as anode of lithium ion batteries. Coat. 2021;11(9):1047. doi:10.3390/coatings11091047
Galashev AY, Vorob'ev AS. First principle modeling of a silicene anode for lithium ion batteries. Electrochim Acta. 2021;378:138143. doi:10.1016/j.electacta.2021.138143
Gevel TA, Zhuk SI, Leonova NM, Leonova AM, Suzdaltsev AV, Zaikov YuP. Electrodeposition of silicon from the KCl–CsCl–K2SiF6 melt. Rus Met (Metally). 2022;2022:958–964. doi:10.1134/S0036029522080237
Baranchugov V, Markevich E, Pollak E, Salitra G, Aurbach D. Amorphous silicon thin films as a high capacity anodes for Li-ion batteries in ionic liquid electrolytes. Electrochem Commun. 2007;9:796–800. doi:10.1016/j.elecom.2006.11.014
Airapetov AA, Vasiliev SV, Kulova TL, Lebedev ME, Metlitskaya AV, Mironenko AA, Nikol’skaya NF, Odinokov VV, Pavlov GYa, Pukhov DE, Rudyi A., Skundin AM. Thin film negative electrode based on silicon composite for lithium-ion batteries. Russ Microelectron. 2016;45:285–291. doi:10.1134/S1063739716030021
Wu JJ, Chen Z, Ma W, Dai Y. Thermodynamic estimation of silicon tetrachloride to trichlorosilane by a low temperature hydrogenation technique. Silicon. 2017;9:69–75. doi:10.1007/s12633-015-9353-0
Fukata N, Oshima T, Tsuruid T, Ito S, Murakami K. Synthesis of silicon nanowires using laser ablation method and their manipulation by electron beam. Sci Techn Adv Mater. 2005;6:628–632. doi:10.1016/j.stam.2005.06.015
Cai Z, Li Y, Tian W. Electrochemical behavior of silicon compound in LiF–NaF–KF–Na2SiF6 molten salt. Ionics. 2011;17:821–826. doi:10.1007/s11581-011-0582-y
Hu Y, Wang X, Xiao J, Hou J, Jiao Sh, Zhu H. Electrochemical behavior of silicon (IV) ion in BaF2–CaF2–SiO2 melts at 1573 K. J Electrochem Soc. 2013;160:81–84. doi:10.1149/2.035303jes
Bieber AL, Massot L, Gibilaro M, Cassayre L, Taxil P, Chamelot P. Silicon electrodeposition in molten fluorides. Electrochim Acta. 2012;62:282–289. doi:10.1016/j.electacta.2011.12.039
Maeda K, Yasuda K, Nohira T, Hagiwara R, Homma T. Silicon electrodeposition in water-soluble KF-KCl molten salt: Investigations on the reduction of Si(IV) ions. J. Electrochem Soc. 2015;162(9):D444–D448. doi:10.1149/2.0441509jes
Zou X, Ji L, Yang X, Lim T, Yu ET, Bard AJ. Electrochemical formation of a p-n junction on thin Film silicon deposited in molten salt. J Amer Chem Soc. 2017;139:16060. doi:10.1021/jacs.7b09090
Zhuk SI, Isakov AV, Apisarov AP, Grishenkova OV, Isaev VA, Vovkotrub EG, Zaikov YuP. Electrodeposition of continuous silicon coatings from the KF-KCl-K2SiF6 melts. J. Electrochem. Soc. 2017;164(8):H5135–H5138. doi:10.1149/2.0171708jes
Dong Y, Slade T, Stolt MJ, Li L, Girard SN, Mai L, Jin S. Low-temperature molten-salt production of silicon nanowires by the electrochemical reduction of CaSiO3. Angew Chemie. 2017;129:14645–14649. doi:10.1002/anie.201707064
Juzeliunas E, Fray DJ. Silicon electrochemistry in molten salts. Chem Reviews. 2020;120:1690. doi:10.1021/acs.chemrev.9b00428
Yu Zh, Wang N, Fang Sh, Qi X, Gao Zh, Yang J, Lu Sh. Pilot-plant production of high-performance silicon nanowires by molten salt electrolysis of silic. Ind Eng Chem Res. 2020;59:1–8. doi:10.1021/acs.iecr.9b04430
Laptev MV, Isakov AV, Grishenkova OV, Vorob'ev AS, Khudorozhkova AO, Akashev LA, Zaikov YuP. Electrodeposition of thin silicon films from the KF-KCl-KI-K2SiF6 melt. J Electrochem Soc. 2020;167(4):042506. doi:10.1149/1945-7111/ab7aec
Hiroki N, Yokoshima T, Momma T, Osaka T. Highly durable SiOC composite anode prepared by electrodeposition for lithium secondary batteries. Energy Env Sci. 2012;5(4):6500–6505. doi:10.1039/C2EE03278C
Plugotarenko NK, Myasoedova TN, Grigoryev MN, Mikhailova TS. Electrochemical deposition of silicon-carbon films: A study on the nucleation and growth mechanism. Nanomater. 2019;9:1754. doi:10.3390/nano9121754
Tao H, Nara H, Yokoshima T, Momma T, Osaka T. Silicon composite thick film electrodeposited on a nickel micro-nanocones hierarchical structured current collector for lithium batteries. J Power Sources. 2013;222:503–509. doi:10.1016/j.jpowsour.2012.09.008
Suzdaltsev A. Silicon Electrodeposition for microelectronics and distributed energy: A mini-review. Electrochem. 2022;3:760–768. doi:10.3390/electrochem3040050
Xin Q, Hang T, Nara H, Yokoshima T, Li M, Osaka T. Electrodeposited three-dimensional porous Si–O–C/Ni thick film as high performance anode for lithium-ion batteries. J Power Sources. 2014;272:794–799. doi:10.1016/j.jpowsour.2014.09.042
Sato T, Toda S, Tachikawa T, Phase diagrams of the Nal-KI and KI-Csl binary systems. Denki Kagaku oyobi Kogyo Butsuri Kagaku. 1987;55(8): 617–620. doi:10.5796/kogyobutsurikagaku.55.617
Khudorozhkova AO, Isakov AV, Kataev AA, Redki AA, Zaykov YP. Density of KF–KCl–KI melts. Rus Met (Metally) 2020;2020:918–924. doi:10.1134/S0036029520080078
Gevel T, Zhuk S, Suzdaltsev AV, Zaikov YuP. Study into the possibility of silicon electrodeposition from a low-fluoride KCl-K2SiF6 melt. Ionics. 2022;28:3537–3545. doi:10.1007/s11581-022-04573-9
Shishkin AV, Shishkin VY, Salyulev AB, Kesikopulos VA, Kholkina AS, Zaikov YP. Electrochemical reduction of uranium dioxide in LiCl–Li2O melt. Atomic Energy. 2021;131(2):78–82. doi:10.1007/s10512-022-00850-y
Casimir A, Zhang H, Ogoke O, Amine J, Wu G. Silicon-based anodes for lithium-ion batteries: Effectiveness of materials synthesis and electrode preparation. Nano Energy. 2016;27:359–376. doi:10.1016/j.nanoen.2016.07.023
Trofimov AA, Leonova AM, Leonova NM, Gevel TA. Electrodeposition of silicon from molten KCl-K2SiF6 for lithium-ion batteries. J Electrochem Soc. 2022;169:020537. doi:10.1149/1945-7111/ac4d6b
DOI: https://doi.org/10.15826/chimtech.2022.9.4.24
Copyright (c) 2022 Rayana K. Abdurakhimova, Michail V. Laptev, Natalia M. Leonova, Anastasia M. Leonova, Alexander S. Schmygalev, Andrey V. Suzdaltsev

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice







