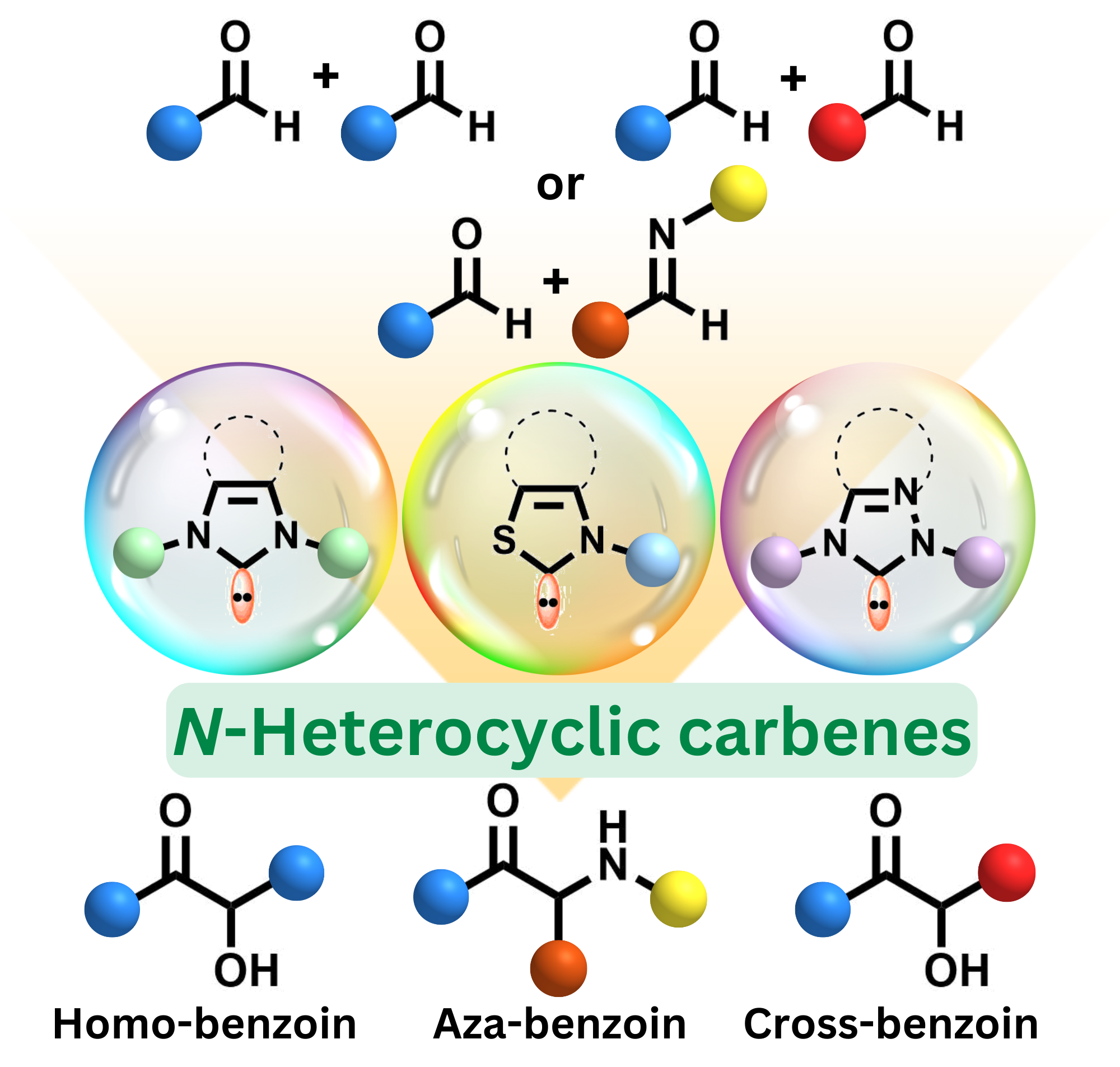
Advancements in N-heterocyclic carbenes (NHCs) catalysis for benzoin reactions: A comprehensive review from past to present
Abstract
Keywords
Full Text:
PDFReferences
Nolan SP, editor. N-Heterocyclic carbenes in synthesis. 1st edition. Weinheim : Chichester: Wiley-VCH; 2006. 319 p. doi:10.1002/9783527609451
Herrmann WA. N-Heterocyclic carbenes: A new concept in organometallic catalysis. Angew Chem Int Ed. 2002;41(8):1290–1309. doi:10.1002/1521-3773(20020415)41:8<1290::AID-ANIE1290>3.0.CO;2-Y
Díez-González S, Nolan SP. Stereoelectronic parameters associated with N-heterocyclic carbene (NHC) ligands: A quest for understanding. Coord Chem Rev. 2007;251(5):874–883. doi:10.1016/j.ccr.2006.10.004
Glorius F. N-Heterocyclic carbenes in transition metal catalysis. Berlin, Heidelberg: Springer Berlin Heidelberg; 2007. (Brown JM, Dixneuf PH, Fürstner A, Hegedus LS, Hofmann P, Knochel P, et al., editors. Topics in Organometallic Chemistry; vol. 21). doi:10.1007/978-3-540-36930-1
Enders D, Balensiefer T. Nucleophilic carbenes in asymmetric organocatalysis. Acc Chem Res. 2004;37(8):534–541. doi:10.1021/ar030050j
Nair V, Bindu S, Sreekumar V. N-Heterocyclic carbenes: Reagents, not just ligands! Angew Chem Int Ed. 2004;43(39):5130–5135. doi:10.1002/anie.200301714
Marion N, Díez-González S, Nolan SP. N-Heterocyclic carbenes as organocatalysts. Angew Chem Int Ed. 2007;46(17):2988–3000. doi:10.1002/anie.200603380
Suzuki Y, Ota S, Fukuta Y, Ueda Y, Sato M. N-Heterocyclic carbene-catalyzed nucleophilic aroylation of fluorobenzenes. J Org Chem. 2008;73(6):2420–2423. doi:10.1021/jo7023569
Lin L, Li Y, Du W, Deng W-P. The NHCs-mediated cross-coupling of aromatic aldehydes with benzyl halides: synthesis of α-aryl ketones. Tetrahedron Lett. 2010;51(27):3571–3574. doi:10.1016/j.tetlet.2010.05.003
Alder RW, Blake ME, Oliva JM. Diaminocarbenes; Calculation of barriers to rotation about Ccarbene−N bonds, barriers to dimerization, proton affinities, and 13C NMR shifts. J Phys Chem A. 1999;103(50):11200–11211. doi:10.1021/jp9934228
Ma Y, Wei S, Wu J, Yang F, Liu B, Lan J, et al. From mono-triazolium salt to bis-triazolium salt: Improvement of the asymmetric intermolecular benzoin condensation. Adv Synth Catal. 2008;350(16):2645–2651. doi:10.1002/adsc.200800371
Enders D, Han J. Synthesis of enantiopure triazolium salts from pyroglutamic acid and their evaluation in the benzoin condensation. Tetrahedron Asymmetry. 2008;19(11):1367–1371. doi:10.1016/j.tetasy.2008.05.017
Breslow R. On the mechanism of thiamine action. IV. Evidence from studies on model systems. J Am Chem Soc. 1958;80(14):3719–3726. doi:10.1021/ja01547a064
Enders D, Kallfass U. An efficient nucleophilic carbene catalyst for the asymmetric benzoin condensation. Angew Chem Int Ed. 2002;41(10):1743–1745. doi:10.1002/1521-3773(20020517)41:10<1743::AID-ANIE1743>3.0.CO;2-Q
Hachisu Y, Bode JW, Suzuki K. Catalytic intramolecular crossed aldehyde−ketone benzoin reactions: A novel synthesis of functionalized preanthraquinones. J Am Chem Soc. 2003;125(28):8432–8433. doi:10.1021/ja035308f
Stetter H, Kuhlmann H. Addition von aliphatischen, heterocyclischen und aromatischen aldehyden an α, β-ungesättigte ketone, nitrile und ester. Chem Ber. 1976;109(8):2890–2896. doi:10.1002/cber.19761090821
Stetter H, Kuhlmann H. The catalyzed nucleophilic addition of aldehydes to electrophilic double bonds. In: Organic Reactions. John Wiley & Sons, Ltd; 2004. p. 407–496. doi:10.1002/0471264180.or040.04
Enders D, Breuer K, Runsink J, Teles JH. The first asymmetric intramolecular stetter reaction. Preliminary communication. Helv Chim Acta. 1996;79(7):1899–1902. doi:10.1002/hlca.19960790712
Kerr MS, Read de Alaniz J, Rovis T. A highly enantioselective catalytic intramolecular stetter reaction. J Am Chem Soc. 2002;124(35):10298–10299. doi:10.1021/ja027411v
Mattson AE, Bharadwaj AR, Scheidt KA. The thiazolium-catalyzed sila-stetter reaction: Conjugate addition of acylsilanes to unsaturated esters and ketones. J Am Chem Soc. 2004;126(8):2314–2315. doi:10.1021/ja0318380
Rose CA, Gundala S, Connon SJ, Zeitler K. Chemoselective crossed acyloin condensations: Catalyst and substrate control. Synthesis. 2010;190–198. doi:10.1055/s-0030-1258363
Wang W, Wang Y, Liu Z, Han Y, Wang C. Study on application performance of oxidized polyethylene wax in powder coatings. Prog Org Coat. 2019;136:105294. doi:10.1016/j.porgcoat.2019.105294
Fan T, Li Z, Cheng B, Li J. Preparation, characterization of PPS micro-porous membranes and their excellent performance in vacuum membrane distillation. J Membr Sci. 2018;556:107–117. doi:10.1016/j.memsci.2018.03.084
Donnelly L, Hardy JG, Gorman SP, Jones DS, Irwin NJ, McCoy CP. Photochemically controlled drug dosing from a polymeric scaffold. Pharm Res. 2017;34(7):1469–1476. doi:10.1007/s11095-017-2164-9
Dharani S, Barakh Ali SF, Afrooz H, Bhattacharya R, Khan MA, Rahman Z. Quality and in-use stability comparison of brand and generics of extended-release phenytoin sodium capsules. J Pharm Sci. 2019;108(5):1808–1817. doi:10.1016/j.xphs.2018.12.022
Rahman Z, Dharani S, Barakh Ali SF, Nutan MTH, Khan MA. Effects of diluents on physical and chemical stability of phenytoin and phenytoin sodium. AAPS PharmSciTech. 2020;21(3):104. doi:10.1208/s12249-020-1639-x
Du TT, Li JF, Min LJ. Green synthesis of phenytoin sodium. Adv Mat Res. 2012;518–523:3917–3920. doi:10.4028/www.scientific.net/AMR.518-523.3917
Kadam A, Jangam S, Oswal R. Application of green chemistry principle in synthesis of phenytoin and its biogical evaluation as anticonvulsant agents. J Chem. 2011;8:S47–S52. doi:10.1155/2011/159430
Safari J, Javadian L. Chitosan decorated Fe3O4 nanoparticles as a magnetic catalyst in the synthesis of phenytoin derivatives. RSC Adv. 2014;4(90):48973–48979. doi:10.1039/C4RA06618A
Tang Y, Cheng Q, Wang S, Zhang J. One-step liquid-phase heterogeneous synthesis of phenytoin using modified calcium oxide as a solid basic catalyst. Monatsh Chem. 2014;145(9):1501–1506. doi:10.1007/s00706-014-1203-z
Konnert L, Reneaud B, de Figueiredo RM, Campagne J-M, Lamaty F, Martinez J, et al. Mechanochemical preparation of hydantoins from amino esters: Application to the synthesis of the antiepileptic drug phenytoin. J Org Chem. 2014;79(21):10132–10142. doi:10.1021/jo5017629
Sachdev D, Dubey A. One step liquid phase heterogeneous synthesis of phenytoin over MgAl calcined hydrotalcites. Catal Commun. 2010;11(13):1063–1067. doi:10.1016/j.catcom.2010.05.004
Three lectures on the preservation of health amongst the personnel of the Japanese navy and army. The Lancet. 1906;167(4317):1451–1455. doi:10.1016/S0140-6736(01)10951-7
Eijkman C. Ein Versuch zur Bekämpfung der Beri-Beri. Archiv f pathol Anat. 1897;149(1):187–194. doi:10.1007/BF01955672
Makino K, Imai T. Bemerkung über die Chemie des antineuritischen Vitamins. Hoppe Seylers Z Physiol Chem. 1936;239(4–6):I–II. doi:10.1515/bchm2.1936.239.4-6.I
Williams RR, Cline JK. Synthesis of vitamin B1. J Am Chem Soc. 1936;58(8):1504–1505. doi:10.1021/ja01299a505
Lohmann K, Schuster P. Untersuchungen uber die cocarboxylase. Biochem Z. 1937;294:188–214.
Ukai T, Tanaka R, Dokawa T. A new catalyst for the acyloin condensation. I. J Pharm Soc Jpn. 1943;63:296–304. doi:10.1248/yakushi1881.63.6_296
Mizuhara S. Action mechanism of vitamin B1. J Jpn Biochem. 1950;22:102–106.
Mizuhara S, Handler P. Mechanism of thiamine-catalyzed reactions. J Am Chem Soc. 1954;76(2):571–573. doi:10.1021/ja01631a071
Lapworth A. XCVI.—Reactions involving the addition of hydrogen cyanide to carbon compounds. J Chem Soc, Trans. 1903;83(0):995–1005. doi:10.1039/CT9038300995
Stetter H, Rämsch RY, Kuhlmann H. Über die präparative nutzung der thiazoliumsalz-katalysierten acyloin- und benzoin-bildung, I. Herstellung von einfachen acyloinen und benzoinen. Synthesis. 1976;1976(11):733–735. doi:10.1055/s-1976-24177
Xu L-W, Gao Y, Yin J-J, Li L, Xia C-G. Efficient and mild benzoin condensation reaction catalyzed by simple 1-N-alkyl-3-methylimidazolium salts. Tetrahedron Lett. 2005;46(32):5317–5320. doi:10.1016/j.tetlet.2005.06.015
Iwamoto K, Hamaya M, Hashimoto N, Kimura H, Suzuki Y, Sato M. Benzoin reaction in water as an aqueous medium catalyzed by benzimidazolium salt. Tetrahedron Lett. 2006;47(40):7175–7177. doi:10.1016/j.tetlet.2006.07.153
Bag S, Vaze VV, Degani MS. Microwave assisted benzoin condensation using thiamine as catalyst. J Chem Res. 2006;2006(4):267–269. doi:10.3184/030823406776894157
Iwamoto K, Kimura H, Oike M, Sato M. Methylene-bridged bis(benzimidazolium) salt as a highly efficient catalyst for the benzoin reaction in aqueous media. Org Biomol Chem. 2008;6(5):912–915. doi:10.1039/B719430G
Hahnvajanawong V, Waengdongbung W, Piekkaew S, Phungpis B, Theramongkol P. Benzoin condensation of aromatic aldehydes catalysed by N,N-dimethylbenzimidazolium iodide and NaOH under green conditions. ScienceAsia. 2013;39(1):50. doi:10.2306/scienceasia1513-1874.2013.39.050
Phungpis B, Hahnvajanawong V, Theramongkol P. Benzoin condensation and stetter reaction catalysed by N,N-dimethylbenzimidazolium iodide in [Bmim][OH]. Orient J Chem. 2014;30(3):933–939. doi:10.13005/ojc/300303
Nicholson WI, Seastram AC, Iqbal SA, Reed-Berendt BG, Morrill LC, Browne DL. N-Heterocyclic carbene acyl anion organocatalysis by ball-milling. ChemSusChem. 2020;13(1):131–135. doi:10.1002/cssc.201902346
Morgan JP, Torres EE, Averill R, Brody AM. Updating the benzoin condensation of benzaldehyde using microwave-assisted organic synthesis and N-heterocyclic carbene catalysis. J Chem Educ. 2023;100(2):986–990. doi:10.1021/acs.jchemed.2c00982
Knight RL, Leeper FJ. Comparison of chiral thiazolium and triazolium salts as asymmetric catalysts for the benzoin condensation. J Chem Soc, Perkin Trans 1. 1998;(12):1891–1894. doi:10.1039/A803635G
Baragwanath L, Rose CA, Zeitler K, Connon SJ. Highly enantioselective benzoin condensation reactions involving a bifunctional protic pentafluorophenyl-substituted triazolium precatalyst. J Org Chem. 2009;74(23):9214–9217. doi:10.1021/jo902018j
Enders D, Breuer K, Teles JH. A novel asymmetric benzoin reaction catalyzed by a chiral triazolium salt. Helv Chim Acta. 1996;79(4):1217–1221. doi:10.1002/hlca.19960790427
Rafiński Z. Enantioselective benzoin condensation catalyzed by spirocyclic terpene-based N-heterocyclic carbenes. Tetrahedron. 2016;72(15):1860–1867. doi:10.1016/j.tet.2016.02.049
Yan J, Sun R, Shi K, Li K, Yang L, Zhong G. N-Heterocyclic carbene-catalyzed asymmetric benzoin reaction in water. J Org Chem. 2018;83(14):7547–7552. doi:10.1021/acs.joc.8b00481
Inokuma T, Hashimoto K, Fujiwara T, Sun C, Kuwano S, Yamada K. Remote electronic effect of chiral N-heterocyclic carbene catalyst on an asymmetric benzoin reaction. Chem Eur J. 2023:e202300858. doi:10.1002/chem.202300858
Garmendia S, Lambert R, Wirotius A-L, Vignolle J, Dove AP, O’Reilly RK, et al. Facile synthesis of reversibly crosslinked poly(ionic liquid)-type gels: Recyclable supports for organocatalysis by N-heterocyclic carbenes. Eur Polym J. 2018;107:82–88. doi:10.1016/j.eurpolymj.2018.07.031
Garmendia S, Dove AP, Taton D, O’Reilly RK. Reversible ionically-crosslinked single chain nanoparticles as bioinspired and recyclable nanoreactors for N-heterocyclic carbene organocatalysis. Polym Chem. 2018;9(43):5286–5294. doi:10.1039/C8PY01293H
Miletto I, Meazza M, Paul G, Cossi M, Gianotti E, Marchese L, et al. Influence of pore size in benzoin condensation of furfural using heterogenized benzimidazole organocatalysts. Chem Eur J. 2022;28(72):e202202771. doi:10.1002/chem.202202771
Ruelens W, Mafakheri F, Lierde V, Smet M. N-Heterocyclic carbene catalysed polymerisation of 2,5-diformylfuran. Org polym mater. 2022;4. doi:10.30564/opmr.v4i2.4953
Matsumoto T, Ohishi M, Inoue S. Selective cross-acyloin condensation catalyzed by thiazolium salt. Formation of 1-hydroxy 2-one from formaldehyde and other aldehydes. J Org Chem. 1985;50(5):603–606. doi:10.1021/jo00205a010
Kuhl N, Glorius F. Direct and efficient N-heterocyclic carbene-catalyzed hydroxymethylation of aldehydes. Chem Commun. 2010;47(1):573–575. doi:10.1039/C0CC02416C
Rose CA, Gundala S, Fagan C-L, Franz JF, Connon SJ, Zeitler K. NHC-catalysed, chemoselective crossed-acyloin reactions. Chem Sci. 2012;3(3):735–740. doi:10.1039/C2SC00622G
Enders D, Henseler A. A direct intermolecular cross-benzoin type reaction: N-heterocyclic carbene-catalyzed coupling of aromatic aldehydes with trifluoromethyl ketones. Adv Synth Catal. 2009;351(11–12):1749–1752. doi:10.1002/adsc.200900247
Enders D, Grossmann A, Fronert J, Raabe G. N-heterocyclic carbene catalysed asymmetric cross-benzoin reactions of heteroaromatic aldehydes with trifluoromethyl ketones. Chem Commun. 2010;46(34):6282–6284. doi:10.1039/C0CC02013C
Jin MY, Kim SM, Han H, Ryu DH, Yang JW. Switching regioselectivity in crossed acyloin condensations between aromatic aldehydes and acetaldehyde by altering N-heterocyclic carbene catalysts. Org Lett. 2011;13(5):880–883. doi:10.1021/ol102937w
Piel I, Pawelczyk MD, Hirano K, Fröhlich R, Glorius F. A family of thiazolium salt derived N-heterocyclic carbenes (NHCs) for organocatalysis: Synthesis, investigation and application in cross-benzoin condensation. Eur J Org Chem. 2011;2011(28):5475–5484. doi:10.1002/ejoc.201100870
Jin MY, Kim SM, Mao H, Ryu DH, Song CE, Yang JW. Chemoselective and repetitive intermolecular cross-acyloin condensation reactions between a variety of aromatic and aliphatic aldehydes using a robust N-heterocyclic carbene catalyst. Org Biomol Chem. 2014;12(10):1547–1550. doi:10.1039/C3OB42486C
Langdon SM, Wilde MMD, Thai K, Gravel M. Chemoselective N-heterocyclic carbene-catalyzed cross-benzoin reactions: importance of the fused ring in triazolium salts. J Am Chem Soc. 2014;136(21):7539–7542. doi:10.1021/ja501772m
Haghshenas P, Gravel M. Chemo- and diastereoselective N-heterocyclic carbene-catalyzed cross-benzoin reactions using N-Boc-α-amino aldehydes. Org Lett. 2016;18(18):4518–4521. doi:10.1021/acs.orglett.6b02123
Delany EG, Connon SJ. Highly chemoselective intermolecular cross-benzoin reactions using an ad hoc designed novel N-heterocyclic carbene catalyst. Org Biomol Chem. 2018;16(5):780–786. doi:10.1039/C7OB03005C
Ji H, Xu J, Ren H. Chemoselective intermolecular cross-benzoin reaction of aliphatic aldehydes and isatins via N-heterocyclic carbene catalysis. Synthesis. 2019;51(10):2191–2197. doi:10.1055/s-0037-1612250
Xu J, Peng J, He C, Ren H. N-Heterocyclic carbene catalyzed chemo- and enantioselective cross-benzoin reaction of aldehydes with isatins. Org Chem Front. 2019;6(2):172–176. doi:10.1039/C8QO01085D
Delany EG, Connon SJ. Enantioselective N-heterocyclic carbene-catalysed intermolecular crossed benzoin condensations: improved catalyst design and the role of in situ racemisation. Org Biomol Chem. 2021;19(1):248–258. doi:10.1039/D0OB02017F
Phungpis B, Worawut K, Keawkumsan P. Ionic liquid 1-butyl-3-methylimidazolium bromide ([Bmim][Br]) acted as both solvent and catalyst for a green reaction of cross benzoin condensation. ASEAN j sci technol. 2021;24(2):91–103. doi:10.55164/ajstr.v24i2.242509
Onodera K, Takashima R, Suzuki Y. Selective synthesis of acylated cross-benzoins from acylals and aldehydes via N-heterocyclic carbene catalysis. Org Lett. 2021;23(11):4197–4202. doi:10.1021/acs.orglett.1c01134
Enders D, Niemeier O, Balensiefer T. Asymmetric intramolecular crossed-benzoin reactions by N-heterocyclic carbene catalysis. Angew Chem Int Ed. 2006;45(9):1463–1467. doi:10.1002/anie.200503885
Takikawa H, Hachisu Y, Bode JW, Suzuki K. Catalytic enantioselective crossed aldehyde–ketone benzoin cyclization. Angew Chem Int Ed. 2006;45(21):3492–3494. doi:10.1002/anie.200600268
Kerr MS, Rovis T. Enantioselective synthesis of quaternary stereocenters via a catalytic asymmetric Stetter reaction. J Am Chem Soc. 2004;126(29):8876–8877. doi:10.1021/ja047644h
Enders D, Niemeier O. Thiazol-2-ylidene catalysis in intramolecular crossed aldehyde-ketone benzoin reactions. Synlett. 2004;2004(12):2111–2114. doi:10.1055/s-2004-831306
Enders D, Niemeier O, Raabe G. Asymmetric synthesis of chromanones via N-heterocyclic carbene catalyzed intramolecular crossed-benzoin reactions. Synlett. 2006;2006(15):2431–2434. doi:10.1055/s-2006-950403
He J, Zheng J, Liu J, She X, Pan X. N-Heterocyclic carbene catalyzed nucleophilic substitution reaction for construction of benzopyrones and benzofuranones. Org Lett. 2006;8(20):4637–4640. doi:10.1021/ol061924f
Kankala S, Edulla R, Modem S, Vadde R, Vasam CS. N-Heterocyclic carbene catalyzed intramolecular crossed aldehyde–ketone benzoin condensation in the chalcone of o-phthalaldehyde: a facile synthesis of naphthalenones. Tetrahedron Lett. 2011;52(29):3828–3831. doi:10.1016/j.tetlet.2011.05.070
Stockton KP, Greatrex BW, Taylor DK. Synthesis of allo- and epi-inositol via the NHC-catalyzed carbocyclization of carbohydrate-derived dialdehydes. J Org Chem. 2014;79(11):5088–5096. doi:10.1021/jo500645z
Kang B, Wang Y, Kuwano S, Yamaoka Y, Takasu K, Yamada K. Site-selective benzoin-type cyclization of unsymmetrical dialdoses catalyzed by N-heterocyclic carbenes for divergent cyclitol synthesis. Chem Commun. 2017;53(32):4469–4472. doi:10.1039/C7CC01191A
Shirke RP, Reddy V, Anand RV, Ramasastry SSV. Furans to benzofurans: Intramolecular cross-benzoin reactions catalysed by N-heterocyclic carbenes. Synthesis. 2016;1865–1871. doi:10.1055/s-0035-1560432
Satyam K, Ramarao J, Suresh S. N-Heterocyclic carbene (NHC)-catalyzed intramolecular benzoin condensation–oxidation. Org Biomol Chem. 2021;19(7):1488–1492. doi:10.1039/D0OB02606A
Liu H, He P, Liao X, Zhou Y, Chen X, Ou W, et al. Stereoselective access to silicon-stereogenic silacycles via the carbene-catalyzed desymmetric benzoin reaction of siladials. ACS Catal. 2022;12(16):9864–9871. doi:10.1021/acscatal.2c02805
Murry JA, Frantz DE, Soheili A, Tillyer R, Grabowski EJJ, Reider PJ. Synthesis of α-amido ketones via organic catalysis: Thiazolium-catalyzed cross-coupling of aldehydes with acylimines. J Am Chem Soc. 2001;123(39):9696–9697. doi:10.1021/ja0165943
Li G-Q, Dai L-X, You S-L. Thiazolium-derived N-heterocyclic carbene-catalyzed cross-coupling of aldehydes with unactivated imines. Chem Commun. 2007;(8):852–854. doi:10.1039/B611646A
Sun F, Ye S. N-Heterocyclic carbene-catalyzed [4 + 1] annulation of phthalaldehyde and imines. Org Biomol Chem. 2011;9(10):3632–3635. doi:10.1039/C1OB05092C
DiRocco DA, Rovis T. Catalytic asymmetric cross-aza-benzoin reactions of aliphatic aldehydes with N-boc-protected imines. Angew Chem Int Ed. 2012;51(24):5904–5906. doi:10.1002/anie.201202442
Uno T, Kobayashi Y, Takemoto Y. N-Heterocyclic carbene-catalyzed direct cross-aza-benzoin reaction: Efficient synthesis of α-amino-β-keto esters. Beilstein J Org Chem. 2012;8:1499–1504. doi:10.3762/bjoc.8.169
Sun Z-X, Cheng Y. N-Heterocyclic carbene-catalyzed cascade annulation reaction of o-vinylarylaldehydes with nitrosoarenes: one-step assembly of functionalized 2,3-benzoxazin-4-ones. Org Biomol Chem. 2012;10(20):4088. doi:10.1039/c2ob25137j
Hahnvajanawong V, Waengdongbung W, Theramongkol P. N,N-Dimethylbenzimidazolium iodide as a green catalyst for cross-coupling of aromatic aldehydeswith unactivated imines. Orient J Chem. 2016;32(1):219–225. doi:10.13005/ojc/320123
Wu J, Zhao C, Wang J. Enantioselective intermolecular enamide–aldehyde cross-coupling catalyzed by chiral N-heterocyclic carbenes. J Am Chem Soc. 2016;138(14):4706–4709. doi:10.1021/jacs.5b13501
Li W, Lv J, Chi YR. N-Heterocyclic carbene catalyzed aza-benzoin reaction for access to α-aminoketone molecules containing benzothiazole fragments. Tetrahedron. 2021;94:132311. doi:10.1016/j.tet.2021.132311
DOI: https://doi.org/10.15826/chimtech.2023.10.2.14
Copyright (c) 2023 Pakin Noppawan, Baramee Phungpis, Kanokkan Worawut

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014–2025
eISSN 2411-1414
Copyright Notice







