H/D exchange studies of methane activation mechanisms in heterogeneous catalysis
Abstract
The development of technologies for the efficient conversion of methane and other light hydrocarbons is becoming vital to the chemical industry. The main technologies for methane conversion are based on solid and liquid catalysts such as metals, oxides and oxide-supported metals. Methane is a highly stable molecule, and the analysis of the catalytic activity of materials with respect to the C-H bond cleavage in methane is of paramount importance for the development of novel methane conversion catalysts. One of the most promising methods for studying methane activation over a catalyst is H/D isotope exchange between the gas and condensed phases. The method provides reliable in situ information on the cleavage of chemical bonds in molecules and allows researchers to elucidate the elementary steps of catalytic methane activation and the stable intermediates involved in the activation process. This paper focuses on the critical analysis of H/D isotope exchange studies of the methane activation mechanism over various metals, oxides, composites, and other catalysts, from the earlier studies to the recent advances in the field. The existing theoretical and experimental approaches to study the H/D exchange between methane and a catalyst are discussed in the paper. A critical analysis of the structure-composition-catalytic activity relationships of the catalysts with respect to methane activation is provided.
Keywords
Full Text:
PDFReferences
Batholomew CH, Carbon Deposition in Steam Reforming and Methanation, Catal. Rev. -Sci. Eng. 24 (1982) 67–112. https://doi.org/10.1080/03602458208079650
Shi Z, Luo JL, Wang S, Sanger AR, et al., Protonic membrane for fuel cell for co-generation of power and ethylene, J. Power Sources 176 (2008) 122–127. https://doi.org/10.1016/j.jpowsour.2007.10.056
Duan C, Kee RJ, Huayang Z, Shang M, et al., Highly durable, coking and sulfur tolerant, fuel-flexible protonic ceramic fuel cells, Nature 557 (2018) 217–235. https://doi.org/10.1038/s41586-018-0082-6
Duan C, Tong J, Shang M, Nikodemski S, et al., Readily processed protonic ceramic fuel cells with high performance at low temperatures, Science 359 (2015) 1321–1326. https://doi.org/10.1126/science.aab3987
Duan C, Kee RJ, Huayang Z, Sullivan N, et al., Highly efficient reversible protonic ceramic electrochemical cells for power generation and fuel production, Nat. Energy 4 (2019) 230–240. https://doi.org/10.1038/s41560-019-0333-2
Shi N, Xue S, Xie Y, Yang Y, et al., Co-generation of electricity and olefin via proton conducting fuel cells using (Pr0.3Sr0.7)0.9Ni0.1Ti0.9O3 catalyst layers, Appl. Catal. B: Environ., 272 (2020) 118973. https://doi.org/10.1016/j.apcatb.2020.118973
Langguth J, Dittmeyer R, Hofmann H., Tomandl G., Studies on oxidative coupling of methane using high-temperature proton-conducting membranes, Appl. Catal. A-Gen., 158 (1997) 287–305. https://doi.org/10.1016/S0926-860X(96)00375-4
Morejudo SH, Zanón R, Escolástico S, Yuste-Tirados et al., Direct conversion of methane to aromatics in a catalytic co-ionic membrane reactor, Science 353 (2016) 563–566. https://doi.org/10.1126/science.aag0274
Nahar G, Dupont V, Hydrogen production from simple alkanes and oxygenated hydrocarbons over ceria–zirconia supported catalysts: Review, Renew. Sust. Energ. Rev. 32 (2014) 777–796. https://doi.org/10.1016/j.rser.2013.12.040
Karakaya C, Kee RJ, Progress in the direct catalytic conversion of methane to fuels and chemicals, Prog. Energ. Combust. 55 (2016) 60–97. https://doi.org/10.1016/j.pecs.2016.04.003
Khoshtinat Nikoo M, Amin NAS, Thermodynamic analysis of carbon dioxide reforming of methane in view of solid carbon formation, Fuel Process. Technol. 92 (2011) 678–691. https://doi.org/10.1016/j.fuproc.2010.11.027
Menon U, Rahman M, Khatib SJ, A critical literature review of the advances in methane dehydroaromatization over multifunctional metal-promoted zeolite catalysts, Appl. Catal. A-Gen. 608 (2020) 117870. https://doi.org/10.1016/j.apcata.2020.117870
Speight JG, The Refinery of the Future. Amsterdam: Elsevier; 2010. 416 p.
Morikawa K, Benedict WS, Taylor HS, The activation of specific bonds in complex molecules at catalytic surfaces. I. The carbon-hydrogen bond in methane and methane-d4, J. Am. Chem. Soc. 58 (1936) 1445–1449. https://doi.org/10.1021/ja01299a040
Raza J, Khoja AH, Anwar M, Saleem F, et al., Methane decomposition for hydrogen production: A comprehensive review on catalyst selection and reactor systems, Renew. Sust. Energ. Rev. 168 (2022) 112774. https://doi.org/10.1016/j.rser.2022.112774
Angeli SD, Monteleone G, Giaconia A, Lemonidou AA, State-of-the-art catalysts for CH4 steam reforming at low temperature, Int. J. Hydrogen Energy 39 (5) (2014) 1979–1997. https://doi.org/10.1016/j.ijhydene.2013.12.001
Kemball C, The Catalytic Exchange of Hydrocarbons with Deuterium, Adv. Catal. 11 (1959) 223–262. https://doi.org/10.1016/S0360-0564(08)60419-8
Frennet A, Chemisorption and exchange with deuterium of methane on metals, Catal. Rev. - Sci. Eng. 10 (1) (1974) 37–68. https://doi.org/10.1080/01614947408079626
Sattler, A. Hydrogen/Deuterium (H/D) Exchange Catalysis in Alkanes, ACS Catal. 8 (2018) 2296–2316. http://dx.doi.org/10.1021/acscatal.7b04201
Rideal EK, A note on a simple molecular mechanism for heterogeneous catalytic reactions, Math. Proc. Cambridge Philos. Soc. 35 (1939) 130–132. https://doi.org/10.1017/S030500410002082X
Horiuti I, Polaniy M, Exchange reactions of hydrogen on metallic catalysts, Farad. Trans. 30 (1934) 1164–1172. https://doi.org/10.1039/TF9343001164
Morikawa K, Benedict WS, Taylor HS, Communications to the editor, J. Am. Chem. Soc. 57 (3) (1935) 590–593. https://doi.org/10.1021/ja01306a063
Wright MM, Taylor HS, The interaction of methane and methane-d4 on nickel and the state of the catalyst surface, J. Can. Res. B. 27 (4) (1949) 303–317. https://doi.org/10.1139/cjr49b-033
Kemball C, Reaction of methane and deuterium on evaporated nickel catalysts, Proc. R. Soc. Lond. A. 207 (1091) (1951) 539–554. https://doi.org/10.1098/rspa.1951.0139
Kramer GJ, Van Santen RA, Emels CA, Nowak AK, Understanding of acid behaviour of zeolites from theory and experiment, Nature 363 (1993) 529–531. https://doi.org/10.1038/363529a0
Evans MW, Bauer N, Beach JY, Mass Spectrometric Study of CH3D. Dissociation Probabilities of C–H and C–D Bonds by Electron Impact, J. Chem. Phys. 14 (1946) 701–707. https://doi.org/10.1063/1.1724089
Schissler DO, Thompson SO, Turkevich J, Behaviour of paraffin hydrocarbons on electron impact, Faraday Discuss. 10 (1951) 46–53. https://doi.org/10.1039/DF9511000046
Mohler FL, Dibeler VH, Quinn E, Redetermination of Mass Spectra of Deuteromethanes, J. Res. Natl. Bur. Stand. 61 (3) (1958) 1711172. http://dx.doi.org/10.6028/jres.061.020
Meija J, Mester Z, D’Ulivo A, Mass spectrometric separation and quantitation of overlapping isotopologues. H2O/HOD/D2O and H2Se/HDSe/D2Se mixtures, J. Am. Soc. Mass. Spectrom. 17 (7) (2006) 1028–1036. https://doi.org/10.1016/j.jasms.2006.02.008
Schoofs B, Martens JA, Jacobs PA, Schoonheydt RA, Kinetics of Hydrogen–Deuterium Exchange Reactions of Methane and Deuterated Acid FAU- and MFI-Type Zeolites, J. Catal. 183 (1999) 355–367. https://doi.org/10.1006/jcat.1999.2401
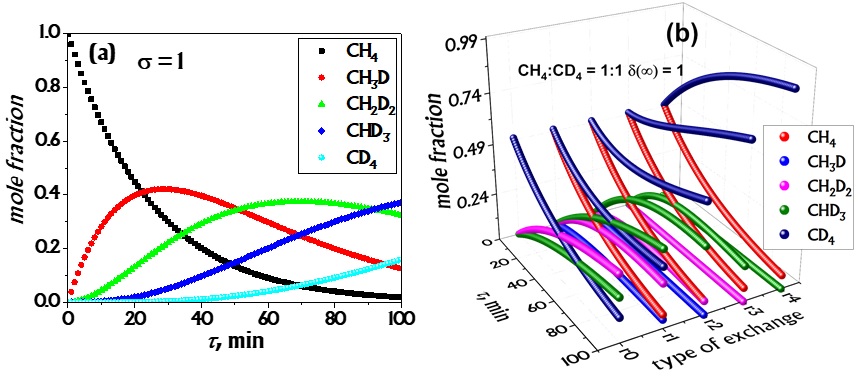
Ananyev MV, Zakharov DM, H/D isotopic exchange between methane and a proton-conducting oxide: theory and experiment, Catal. Sci. Technol. 10 (2020) 3561–3571. https://doi.org/10.1039/C9CY02566A
Schuurman Y, Mirodatos C, Uses of transition kinetics for methane activation studies, Appl. Cat. A-Gen. 151 (1997) 305–331. https://doi.org/10.1016/S0926-860X(97)00031-8
Osawa T, Hamano Y, Saga S, Takayasu A, Hydrogen–deuterium exchange of methane on nickel surface prepared by the reduction of nickel oxide, J. Molec. Catal. A. Chem. 298 (2009) 111–114. https://doi.org/10.1016/j.molcata.2008.09.030
Quanzhi L, Amenomiya Y, Exchange reaction of methane on some oxide catalyst, Appl. Catal. 23 (1) (1986) 173–182. https://doi.org/10.1016/S0166-9834(00)81460-8
Walspurger S, Goeppert A, Haouas M, Sommer J, H/D isotope exchange between methane and magic acid (HSO3F–SbF5): an in situ NMR study, New J. Chem. 28 (2004) 266–269. https://doi.org/10.1039/B309362J
Stepanov AG, Arzumanov SS, Gabrienko AA, Tokatrev AV, et al., Zn-promoted hydrogen exchange for methane and ethane on Zn/H-BEA zeolite: In situ 1H MAS NMR kinetic study, J. Catal. 253 (2008) 11–21. https://doi.org/10.1016/j.jcat.2007.11.002
Klier K, Novakova J, Jiru P, Exchange Reactions of Oxygen between Oxygen Molecules and Solid Oxides, J. Catal. 2 (1963) 479–484. https://doi.org/10.1016/0021-9517(63)90003-4
Boreskov GK, Muzykantov VS, Investigation of oxide-type oxidation catalysts by reactions of oxygen isotopic exchange, Ann. N. Y. Acad. Sci., 213 (1973) 137−170. https://doi.org/10.1111/j.1749-6632.1973.tb51065.x
Den Otter MW, Boukamp BA, Bouwmeester HJM, Theory of oxygen isotope exchange, Solid State Ionics, 139 (2001) 89–94. https://doi.org/10.1016/S0167-2738(00)00801-8
Lacombe S, Holmen A, Wolf EE, Ducarme V, et al., Isotopic exchange and volumetric studies on methane activation over rare-earth oxides, Stud. Surf. Sci. Catal. 81 (1994) 211–216. https://doi.org/10.1016/S0167-2991(08)63867-7
Kemball C, Catalysis on evaporated metal films. II. The efficiency of different metals for the reaction between methane and deuterium, Proc. R. Soc. Lond. A. 217 (1130) (1953) 376–389. https://doi.org/10.1098/rspa.1953.0069
Anderson JR, Kemball C, Catalysis on evaporated metal films. III. The efficiency of different metals for the reaction between ethane and deuterium, Proc. R. Soc. Lond. A. 223 (1154) (1954) 361–367. https://doi.org/10.1098/rspa.1954.0121
McKay HAC, Kinetics of Exchange reactions, Nature 142 (1938) 997. https://doi.org/10.1038/142997b0
Miyahara K, Exchange reaction between methane and deuterium, J. Res. Inst. Catal., Hokkaido Univ. 4 (3) (1957) 177–184. https://eprints.lib.hokudai.ac.jp/dspace/handle/2115/24646
Bolder H, Dallinga G, Kloosterziel H, Kinetics and distribution in hydrogen isotope exchange and redistribution, J. Catal. 3 (3) (1964) 312–340. https://doi.org/10.1016/0021-9517(64)90037-5
Shestov AA. Izotopnaya kinetica obmena I mehanismy heterogenno-kataliticheskoy activacsii metana I etena [dissertation]. Novosibirsk (Russia): Boreskov institute of catalysis; 1993. 243.
Muzykantov VS, Shestov AA, Ehwald H, Mechanisms of catalytic activation of methane and ethylene on magnesia surface elucidated by isotopic kinetics, Catal. Today, 24 (1995) 243–422. https://doi.org/10.1016/0920-5861(95)00032-B
Muzykantov VS, Shestov AA, Kinetic equations for the redistribution of isotopic molecules due to reversible dissociation. Homoexchange of methane, React. Kinet, Catal. Lett. 32 (1986) 307–312. https://doi.org/10.1007/BF02068328
Muzykantov VS, Shestov AA, Redistribution kinetics of isotope molecules due to reversible bimolecular reactions with several atomic channels, React. Kinet, Catal. Lett. 33 (1987) 197–202. https://doi.org/10.1007/BF02066723
Muzykantov VS, Distribution and transfer of atoms by elementary, React. Kinet. Catal. Lett. 13 (4) (1980) 419–424. https://doi.org/10.1007/BF02065710
Muzykantov VS, Kinetic equations of isotope redistribution in an elementary reaction, React. Kinet. Catal. Lett. 14 (1) (1980) 113–118. https://doi.org/10.1007/BF02061274
Muzykantov VS, Kinetic equations of isotope transfer by elementary reaction, React. Kinet. Catal. Lett. 14 (2) (1980) 161–167. https://doi.org/10.1007/BF02061281
Muzykantov VS, Kinetics of isotopic exchange due to one elementary reaction, React. Kinet. Catal. Lett. 14 (3) (1980) 311–316. https://doi.org/10.1007/BF02073497
Zakharov DM, Zhuravlev NA, Denisova TA, Belozerov AS et al., Catalytic methane activation over La1−xSrxScO3−α proton-conducting oxide surface: A comprehensive study, J. Catal. 394 (2021) 67–82. https://doi.org/10.1016/j.jcat.2020.12.011
Shi T, Sreedhar D, Zeng L, Chen A, Recent advances in catalyst design for the electrochemical and photoelectrochemical conversion of methane to value-added products, Eletcrochem. Commun. 135 (2022) 107220. https://doi.org/10.1016/j.elecom.2022.107220
McKee DW, Norton FJ, Catalytic Exchange of Methane and Deuterium on Platinum, Ruthenium, and Platinum-Ruthenium Alloys, J. Phys. Chem-US. 68 (3) (1964) 481–489. https://doi.org/10.1021/j100785a008
McKee DW, Norton FJ, Catalytic activity of noble metal alloys. Methane-deuterium exchange and propane cracking on platinum-palladium and palladium-rhodium alloys, J. Catal. 3 (1964) 252–267. https://doi.org/10.1016/0021-9517(64)90172-1
McKee DW, Norton FJ, Methane-Deuterium Exchange over Platinum-Rhodium Alloys, J. Catal. 4 (1965) 510–517. https://doi.org/10.1016/0021-9517(65)90056-4
McKee DW, Catalytic Exchange of Methane with Deuterium on Palladium-Gold Alloys, J. Phys. Chem. US. 70 (2) (1966) 525–530. https://doi.org/10.1021/j100874a034
Guszi L, Karpiski Z, Catalytic Reactions of Hydrocarbons over Pt-Pd Alloys II. Deuterium Exchange of Methane and Ethane over Pt-Pd Alloy Films. Surface Composition of Pt-Pd Alloy System, J. Catal. 56 (1979) 438–444. https://doi.org/10.1016/0021-9517(79)90134-9
Anderson JR, Macdonald RJ, Relation between catalytic properties and structure of metal films. I. Deuterium exchange of methane, ethane and propane over nickel, J. Catal. 13 (4) (1969) 345–359. https://doi.org/10.1016/0021-9517(69)90443-6
Van Der Lugt WThAM, Ros P, Retention and inversion in bimolecular substitution reactions of methane, Chem. Phys. Lett. 4 (6) (1969) 389–392. https://doi.org/10.1016/0009-2614(69)80271-X
Mulder JJC, Wright JS, The electronic structure and stability of CH+5 and CH−5, Chem. Phys. Lett. 5 (8) (1970) 445–449. https://doi.org/10.1016/0009-2614(70)85103-X
Frennet A, Collation between the catalytic! activity of metals and the surface states resulting from the reaction of the gaseous reactants and the metal, Surf. Sci. 18 (1969) 80–96. https://doi.org/10.1016/0039-6028(69)90268-4
Zakharov DM, Ananyev MV, Methane dissociation mechanism on Ni–La1-xSrxScO3-δ cermet for proton ceramic electrochemical devices, Int. J. Hydrogen Energy 47 (38) (2022) 16824–16839. https://doi.org/10.1016/j.ijhydene.2022.03.171
Ananyev MV, Porotnikova NM, Eremin VA, Kurumchin EKh, Interaction of O2 with LSM–YSZ Composite Materials and Oxygen Spillover, ACS Catal. 11 (7) (2021) 4247–4262. https://doi.org/10.1021/acscatal.0c04558
Kopylets VI, Heats of adsorption of hydrogen on metals, Mater. Sci. 35 (1999) 438–441. https://doi.org/10.1007/BF02355490
Holleck GL, Diffusion and solubility of hydrogen in palladium and palladium silver alloys, J. Phys. Chem-US. 74 (3) (1970) 503–511. https://doi.org/10.1021/j100698a005
Lewis FA, Solubility of hydrogen in metals, Pure Appl. Chem. 62 (11) (1990) 2091–2096. https://doi.org/10.1351/pac199062112091
Kolachev BA, Vodorodnaya hrupkost’ csvetnih metallov [hydrogen brittleness of nonferrous metals. Moscow: Metalurgia; 1966. 250 p. Russian
Debe MK, Electrocatalyst approaches and challenges for automotive fuel cells, Nature 486 (2012) 43–51. https://doi.org/10.1038/nature11115
Vignarooban K, Lin J, Arvay A, Kolli S, et al., Nano-electrocatalyst materials for low temperature fuel cells: A review, Chin. J. Catal. 36 (2015) 458–472. https://doi.org/10.1016/S1872-2067(14)60175-3
Fergus JW, Oxide anode materials for solid oxide fuel cells, Solid State Ionic, 177 (17-18) (2006) 1529–1541. https://doi.org/10.1016/j.ssi.2006.07.012
Sood K, Tiwary P, Dhall S, Basu S, Understanding electrocatalytic activity of titanium and samarium doped ceria as anode material for solid oxide fuel cells, J. Electroanal. Chem. 925 (2022) 116902. https://doi.org/10.1016/j.jelechem.2022.116902
Kim S, Avila-Paredes HJ, Wang S, Chen C-T, et al., On the conduction pathway for protons in nanocrystalline yttria-stabilized zirconia, Phys. Chem. Chem. Phys. 11 (2009) 3035−3038. https://doi.org/10.1039/B901623F.
Scherrer B, Schlupp MVF, Stender D, Martynczuk J, et al., On Proton Conductivity in Porous and Dense Yttria Stabilized Zirconia at Low Temperature, Adv. Funct. Mater., 23 (2013) 1957–1964. https://doi.org/10.1002/adfm.201202020.
Avila-Paredes HJ, Zhao J, Wang S, Pietrowski M, et al. Protonic conductivity of nano-structured yttria-stabilized zirconia: dependence on grain size, J. Mater. Chem., 20 (2010) 990–994. https://doi.org/10.1039/B919100C.
Miyoshi S, Akao Y, Kuwata N, Kawamura J, et al., Low-Temperature Protonic Conduction Based on Surface Protonics: An Example of Nanostructured Yttria-Doped Zirconia, Chem. Mater., 26 (2014) 5194–5200. https://doi.org/10.1021/cm5012923
Kreuer KD, Adams St, Münch W, Fuchs A, et al., Proton conducting alkaline earth zirconates and titanates for high drain electrochemical applications, Solid State Ionics. 145 (1-4) (2001) 295–306. https://doi.org/10.1016/S0167-2738(01)00953-5
Larson JG, Hall WK, VII. Studies of hydrogen held by solids. The exchange of the hydroxyl group of alumina and silica-alumina catalyst with deuterated methane, J. Phys. Chem-US 69 (1965) 3080–3089. https://doi.org/10.1021/j100893a044
Perri JB, Infrared and gravimetric study of the surface hydration of γ-Al2O3, J. Phys. Chem-US. 69 (1) (1965) 211–219. https://doi.org/10.1021/j100885a032
Perri JB, Hannan RB, Surface hydroxyl groups on γ-Alumina, J. Phys. Chem-US. 64 (10) (1960) 1526–1530. https://doi.org/10.1021/j100839a044
Brönsted JN, Einige Bemerkungen über den Begriff der Säuren und Basen, Recl. Trav. Chim. Pays-Bas. 42 (8) (1923) 718–728. http://dx.doi.org/10.1002/recl.19230420815
Lowry TM, The uniqueness of hydrogen, J. Soc. Chem. Ind., London. 42 (3) (1923) 43–47. https://doi.org/10.1002/jctb.5000420302
Engelhardt J, Valyon J, H/D exchange between CD4 and the OH-groups of H-zeolites and γ-Al2O3, React. Kinet, Catal. Lett. 74 (2001) 217–224. https://doi.org/10.1023/A:1017972624428
Bjørheim TS, Hoedi MF, Merkle R, Kotomin EA, et al., Proton, Hydroxide Ion, and Oxide Ion Affinities of Closed-Shell Oxides: Importance for the Hydration Reaction and Correlation to Electronic Structure, J. Phys. Chem. C. 124 (2) (2020) 1277–1284. https://doi.org/10.1021/acs.jpcc.9b07570
Utiyama M, Hattori H, Tanabe K, Exchange Reaction of Methane with Deuterium over Solid Base Catalysts, J. Catal. 53 (1978) 237–242. https://doi.org/10.1016/0021-9517(78)90071-4
Hargreaves JSJ, Hatchings GJ, Joyner RW, Taylor SH, A study of the methane–deuterium exchange reaction over a range of metal oxides, Appl. Catal. A-Gen. 227 (2002) 191–200. https://doi.org/10.1016/S0926-860X(01)00935-8
Bird R, Kemball C, Frank Leach H, Reactions of Alkanes with Deuterium on Lanthana in the Temperature Range 570 to 720 K, J. Catal. 107 (1987) 424–433. https://doi.org/10.1016/0021-9517(87)90306-X
Kalman J, Guczi L, Hydrogen-Deuterium Exchange in Saturated Hydrocarbons on α-Chromia Catalyst, J. Catal. 47 (1977) 371–383. https://doi.org/10.1016/0021-9517(77)90186-5
Robertson PJ, Scurrel MS, Kemball C, Exchange of alkanes with deuterium over γ-alumina. A Brønsted linear free energy relationship, Farad. Trans. 71 (1975) 903–912. https://doi.org/10.1039/F19757100903
Parravano G, Hammel EF, Taylor HS, The exchange reaction between methane and deuteromethanes on silica-alumina cracking catalysts, J. Am. Chem. Soc. 70 (1948) 2269–2270. https://doi.org/10.1021/ja01186a083
Kramer GJ, van Santen RA, An ab initio study of D/H exchange between CD4 and the H-Forms of zeolites FAU and MFI, J. Am. Chem. Soc. 117 (1995) 1766–1776. https://doi.org/10.1021/ja00111a014
Blaszkowski SR, Jansen APJ, Nascimento MAC, van Santen RA, Density functional theory calculations of the transition states for hydrogen exchange and dehydrogenation of methane by a Bronsted zeolitic proton, J. Phys. Chem-US. 98 (1994) 12938–12944. https://doi.org/10.1021/j100100a021
Lee B, Kondo JN, Wakabayashi F, Domen K, Infrared study of H/D isotope exchange reaction of alkanes and Bronsted acidic hydroxyl groups of FER zeolite, Catal. Lett. 59 (1999) 51–54. https://doi.org/10.1023/A:1019047916347
Vollmer JM, Truong TN, Mechanisms of hydrogen exchange of methane with H-Zeolite Y: an ab initio embedded cluster study, J. Phys. Chem. B. 104 (2000) 6308–6312. https://doi.org/10.1021/jp0008445
Zheng XA, Blowers P, Computational study of alkane hydrogen-exchange reactions on zeolites, Mol. Catal. A-Chem. 242 (2005) 18–25. https://doi.org/10.1021/jp990555k
Keith Hall W, Leftin HP, Cheselske FJ, O’Reilly DE, Studies of Hydrogen held by solids IV. Deuterium Exchange and NMR investigations of Silica, Alumina, and Silica-alumina catalysts, J. Catal. 2 (1963) 506–517. https://doi.org/10.1016/0021-9517(63)90006-X
Haag WO, Lago RM, Weisz PB, The active site of acidic aluminosilicate catalysts, Nature 309 (1984) 589–591. https://doi.org/10.1038/309589a0
Vaylon J, Engelhardt J, Kallo D, Hegedus M, The activation of CD 4 for H/D exchange over H-zeolites, Catal. Lett. 82 (1-2) (2002) 29–35. https://doi.org/10.1023/A:1020579705752
Vdovin GK, Kurumchin EKh, High-temperature proton conductors based on strontium and barium cerates: The content, interphase exchange, and diffusion of hydrogen, Russ. J. Electrochem. 40 (2004) 403–409. https://doi.org/10.1023/B:RUEL.0000023931.90282.e1
Ananyev MV, Farlenkov AS, Kurumchin EKh, Isotopic exchange between hydrogen from the gas phase and proton-conducting oxides: Theory and experiment, Int. J. Hydrogen Energy 43 (29) (2018) 13373–13382. https://doi.org/10.1016/j.ijhydene.2018.05.150
Shin HH, McIntosh S, On the H2/D2 isotopic exchange rate of proton conducting barium cerates and zirconates, J. Mater. Chem. A. 1 (2013) 7639. https://doi.org/10.1039/C3TA10740J
Hancke R, Li Z, Haugsrud R, Hydrogen surface exchange on proton conducting oxides studied by gas phase analysis with mass spectrometry, J. Membr. Sci. 439 (2013) 68–77. https://doi.org/10.1016/j.memsci.2013.03.049
Farlenkov AS, Zhuravlev NA, Denisova TA, Ananyev MV, Interaction of O2, H2O and H2 with proton-conducting oxides based on lanthanum scandates, Int. J. Hydrogen Energy 44 (2019) 26419–26427. https://doi.org/10.1016/j.ijhydene.2019.08.171
Bouwmeester HJM, Song C, Zhu J, Yi J, et al., A novel pulse isotopic exchange technique for rapid determination of the oxygen surface exchange rate of oxide ion conductors, Phys. Chem. Chem. Phys. 11 (2009) 9640–9643. https://doi.org/10.1039/B912712G
Ananyev MV, Tropin ES, Eremin VA, Farlenkov AS, et al., Oxygen isotope exchange in La2NiO4±δ, Phys. Chem. Chem. Phys. 18 (2016) 9102–9111. https://doi.org/10.1039/C5CP05984D
Singhal SC, Kendall K, High temperature solid oxide fuel cells: Fundamentals, Design and Applications, Amsterdam: Elsevier; 1985. 406 p.
Boudart M, Vannice MA, Benson JE, Adlineation, Portholes and Spillover, Z. Physik. Chem. NeneFolge. 64 (1969) 171–177. https://doi.org/10.1524/zpch.1969.64.1_4.171
Fukunaga H, Ihara M, Sakaki K, Yamada K, The relationship between overpotential and the three phase boundary length, Solid State Ionics 76-88 (1996) 1179–1185. https://doi.org/10.1016/0167-2738(96)00284-6
Sen B, Falconer JL, Spillover sites on a 18% NiAl2O3 catalyst, J. Catal. 117 (1989) 404–415. https://doi.org/10.1016/0021-9517(89)90351-5
Verkerk MJ, Hammink MW, Burggraaf AJ, Oxygen transfer on substituted ZrO2, Bi2O3 and CeO2 electrolytes with platinum electrodes, J. Electrochem. Soc. 130 (1983) 70–78. https://doi.org/10.1149/1.2119687
Fabry P, Kleitz M, Influence of the metal and the electrolyte composition on the characteristics of the oxygen electrode reaction on solid oxygen electrolyte, Electroanal. Chem. 57 (1974) 165–177. https://doi.org/10.1016/S0022-0728(74)80020-3
Bieberle A, Meier LP, Gauckler LJ, The Electrochemistry of Ni Pattern Anodes Used as Solid Oxide Fuel Cell Model Electrodes, J. Electrochem. Soc. 148 (2001) A646–A656. https://doi.org/10.1149/1.1372219
Vogler M, Bieberle-Hütter A, Gauckler L, Warnatz J, et al. Modelling Study of Surface Reactions, Diffusion, and Spillover at a Ni-YSZ Patterned Anode, J. Electrochem. Soc. 156 (2009) B663–B672. http://dx.doi.org/10.1149/1.3095477
Williford RE, Chick A, Maupin GD, Simner SP, Diffusion Limitations in the Porous Anodes of SOFCs, J. Electrochem. Soc. 150 (2003) A1067–A1072. http://dx.doi.org/10.1149/1.1586300
Bhatia B, Sholl DS, Chemisorption and diffusion of hydrogen on surface and subsurface sites of flat and stepped nickel surfaces, J. Chem. Phys. 122 (2005) 204707. http://dx.doi.org/10.1063/1.1902943
Weng MH, Chen HT, Wang YC, Ju SP, et al. Kinetics and Mechanisms for the Adsorption, Dissociation, and Diffusion of Hydrogen in Ni and Ni-YSZ Slabs: A DFT, Langmuir. 28 (2012) 5596–5605. http://dx.doi.org/10.1021/la300305m
Cece JM, Gonzalez RD, Studies of Supported Metal Catalysts II. The Effect of Dispersion on Methane-Deuterium Exchange Over Supported Nickel, J. Catal. 28 (1973) 260–264. https://doi.org/10.1016/0021-9517(73)90009-2
Leach HF, Mirodatos C, Whan DA, The Exchange of Methane, Ethane, and Propane with Deuterium on Silica-Supported Nickel Catalyst, J. Catal. 63 (1980) 138–151. https://doi.org/10.1016/0021-9517(80)90067-6
Dalmon JA, Mirodatos C, Exchange with deuterium of methane on nickel catalysts, J. Mol. Catal. 25 (1984) 161–172. https://doi.org/10.1016/0304-5102(84)80040-1
Oliver JA, Kemball C, Brown R, Jamieson EW, Exchange Reactions of Hydrocarbons on Silica-supported Rh-Pt Bimetallic Catalyst, Farad. Trans. 81 (1985) 1871–1881. https://doi.org/10.1039/F19858101871
Miura H, Ushikubo Y, Sugiyama K, Matsuda T, Effect of the surface composition of silica supported Pt-Ru bimetallic catalysts on methane-deuterium exchange reaction, React. Kinet. Catal. Lett. 32 (1986) 487–498. https://doi.org/10.1007/BF02068356
Garden D, Kemball C, Whan DA, Reactions of Hydrocarbons on Alumina-supported Pt-Ir Bimetallic Catalysts, Farad. Trans. 82 (1986) 3113–3123. https://doi.org/10.1039/F19868203113
Brown R, Kemball C, Reactions of Hydrocarbons over Ru/SiO2: Exchange with Deuterium and the Onset of Hydrogenolysis, Farad. Trans. 89 (1993) 585–594. https://doi.org/10.1039/FT9938900585
Da Costa Faro A, Kemball C, Influence of Strong Metal-Support Interaction on Exchange with Deuterium and other Reactions of Hydrocarbons Part 1.-Studies with Rh/TiO2, and Rh/SiO2, Farad. Trans. 91 (1995) 741–748. https://doi.org/10.1039/FT9969200281
Otsuka K, Kobayashi S, Takenaka S, Hydrogen–Deuterium Exchange Studies on the Decomposition of Methane over Ni/SiO2, J. Catal. 200 (2001) 4–9. https://doi.org/10.1006/jcat.2001.3179
Mirich A, Miller TH, Klotz E, Mattson B, Heterogeneous Catalysis: Deuterium Exchange Reactions of Hydrogen and Methane, Chem. Educ. 92 (2015) 2087–2093. https://doi.org/10.1021/acs.jchemed.5b00360
Kroll VCH, Swaan HM, Lacombe S, Mirodatos C, Methane reforming reaction with carbon dioxide over Ni/SiO2 catalyst II. A Mechanistic Study, J. Catal. 164 (1997) 387–398. https://doi.org/10.1006/jcat.1996.0395
Otsuka K, Production of Hydrogen through Decomposition of Methane with Ni-supported Catalysts, Chem. Lett. 28 (1999) 1179–1180. https://doi.org/10.1246/cl.1999.1179
Otsuka K, Kobayashi S, Takenaka S, Decomposition and regeneration of methane in the absence and the presence of a hydrogen-absorbing alloy CaNi5, Appl. Catal. A-Gen. 190 (2000) 261–268. https://doi.org/10.1016/S0926-860X(99)00324-5
Khodakov A, Barbouth N, Berthler Y, Oudar J, et al. Effect of Pt Particle Size on H/D Exchange of Methane over Alumina and Zeolite-supported Catalysts, Farad. Trans. 91 (1995) 569–573. https://doi.org/10.1039/FT9959100569
Wei J, Iglesia E, Reaction Pathways and Site Requirements for the Activation and Chemical Conversion of Methane on Ru-Based Catalysts, J. Phys. Chem. B. 108 (2004) 7253–7262. https://doi.org/10.1021/jp030783l
Wen B, Sachtler WMH, H/D exchange of methane over transition metal/MFI catalysts, Appl. Catal. A-Gen. 229 (2002) 11–22. https://doi.org/10.1016/S0926-860X(02)00012-1
Arzumanov SS, Moroz IB, Freude D, Haase J, et al., Methane Activation on In-Modified ZSM‑5 Zeolite. H/D Hydrogen Exchange of the Alkane with Brønsted Acid Sites, J. Phys. Chem. C. 118 (2014) 14427–14432. https://doi.org/10.1021/jp5037316
Arzumanov SS, Gabrienko AA, Freude D, Stepanov AG, Competitive pathways of methane activation on Zn2+-modified ZSM-5 zeolite: H/D hydrogen exchange with Brønsted acid sites versus dissociative adsorption to form Zn-methyl species, Catal. Sci. Technol. 6 (2016) 6381. https://doi.org/10.1039/c6cy00878j
Gabrienko AA, Arzumanov SS, Toktarev AV, Freude D, et al., Hydrogen H/D Exchange and Activation of C1-n-C4 Alkanes on Ga-Modified Zeolite BEA Studied with 1 H Magic Angle SpinningNuclear Magnetic Resonance in Situ, J. Phys. Chem. C 115 (2011) 13877–13886. https://dx.doi.org/10.1021/jp204398r
Gabrienko AA, Arzumanov SS, Moroz IB, Toktarev AV, et al., Methane Activation and Transformation on Ag/H-ZSM 5 Zeolite Studied with Solid-State NMR, J. Phys. Chem. C 117 (2013) 7690–7702. https://dx.doi.org/10.1021/jp4006795
Gabrienko AA, Arzumanov SS, Toktarev AV, Danilova IG, et al., Different Efficiency of Zn2+ and ZnO Species for Methane Activation on Zn-Modified Zeolite, ACS Catal. 7 (3) (2017) 1818–1830. https://doi.org/10.1021/acscatal.6b03036
Gabrienko AA, Kolganov AA, Arzumanov SS, Yashnik SA, et al., Effect of Copper State in Cu/H-ZSM 5 on Methane Activation by Brønsted Acid Sites, Studied by 1H MAS NMR In Situ Monitoring the H/D Hydrogen Exchange of the Alkane with Brønsted Acid Sites, J. Phys. Chem. C 125 (2021) 2182–2193. https://dx.doi.org/10.1021/acs.jpcc.0c10261?ref=pdf
Zakharov DM, Tropin ES, Osinkin DA, Farlenkov AS, et al., H/D isotopic exchange and electrochemical kinetics of hydrogen oxidation on Ni-cermets with oxygen-ionic and protonic electrolytes, J. Power Sources 517 (2022) 230708. https://doi.org/10.1016/j.jpowsour.2021.230708
Naito S, Takada A, Tokizawa S, Miyao T, Mechanistic study on the methane activation over various supported molybdenum carbide catalysts with isotopic tracer methods, Appl. Catal. A Gen. 289 (2005) 22–27. https://doi.org/10.1016/j.apcata.2005.04.011
Brush A, Mullen GM, Dupre R, Kota S, et al., Evidence of methane adsorption over Mo2C involving single C–H bond dissociation instead of facile carbon exchange, React. Chem. Eng. 1 (2016) 667. https://doi.org/10.1039/c6re00141f
Hua W, Goeppert A, Sommer J, H/D Exchange and Isomerization of Small Alkanes over Unpromoted and Al2O3-Promoted SO2−4/ZrO2 Catalysts, J. Catal. 197 (2001) 406–413. https://doi.org/10.1006/jcat.2000.3106
Hua W, Goeppert A, Sommer J, Methane activation in the presence of Al2O3 -promoted sulfated zirconia, Appl. Catal. A Gen. 219 (2001) 201–207. https://doi.org/10.1016/S0926-860X(01)00686-X
Jentoft RA, Gates BC, H–D exchange between CD 4 and solid acids: AlCl3/sulfonic acid resin, promoted and unpromoted sulfated zirconia, and zeolite HZSM-5, Catal. Lett, 72 (3-4) (2001) 129–133. https://doi.org/10.1023/A:1009075809619
Hanada H, Baba T, Ono Y, H–D Exchange between methane and deuteriated potassium amide supported on alumina, J. Chem. Soc., Faraday T rans., 94 (3) (1998) 451–454. https://doi.org/10.1039/A706059I
Olah GA, Klopman G, Schlosberg RH, Chemistry in super acids. I. Hydrogen exchange and polycondensation of methane and alkanes in FSO3H-SbF5 ("magic acid") solution. Protonation of alkanes and the intermediacy of CH5+ and related hydrocarbon ions. The high chemical reactivity of "paraffins" in ionic solution reactions, J. Am. Chem. Soc. 90 (10) (1968) 2726–2727. https://doi.org/10.1021/ja01012a066
Olah GA, Klopman G, Schlosberg RH, Super acids. III. Protonation of alkanes and intermediacy of alkanonium ions, pentacoordinated carbon cations of CH5+ type. Hydrogen exchange, protolytic cleavage, hydrogen abstraction; polycondensation of methane, ethane, 2,2-dimethylpropane and 2,2,3,3-tetramethylbutane in FSO3H-SbF5, J. Am. Chem. Soc. 91 (12) (1969) 3261–3268. https://doi.org/10.1021/ja01040a029
Hogeveen H, Gaasbeek CJ, Electrophilic substitution at methane: Short communication, RECUEL 87 (1968) 319–320. https://doi.org/10.1002/recl.19680870311
Goeppert A, Diner P, Ahlberg P, Sommer J, Methane Activation and Oxidation in Sulfuric Acid, Chem. Eur. J. 8 (14) (2002) 3277–3283. https://doi.org/10.1002/1521-3765(20020715)8:14%3C3277::AID-CHEM3277%3E3.0.CO;2-5
DOI: https://doi.org/10.15826/elmattech.2023.2.014
Copyright (c) 2023 Dmitriy M. Zakharov

This work is licensed under a Creative Commons Attribution 4.0 International License.

