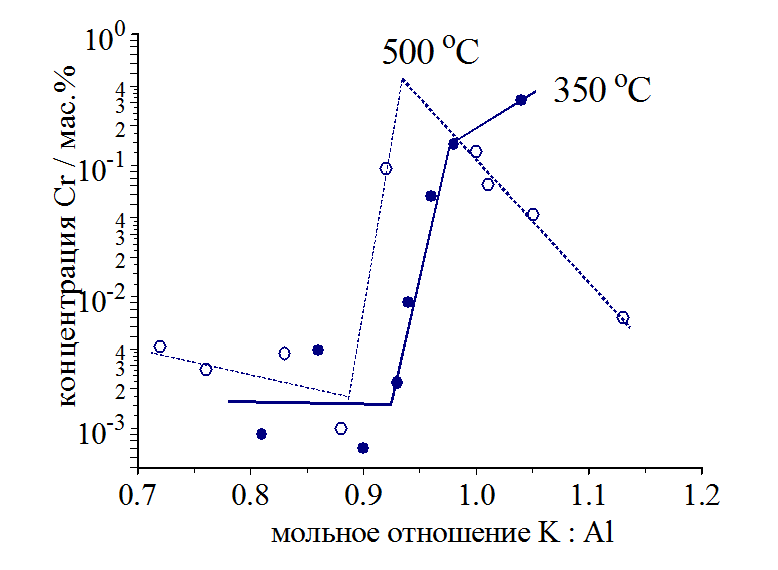
Determination of solubility of chlorides of d-elements in chloroaluminate melts
Abstract
Keywords
Full Text:
PDF (RUSSIAN) (Русский)References
Gilbert B, Osteryoung RA. Electrochemistry of Ni(II) and the behavior of oxide ions in chloroaluminate melts. J Am Chem Soc. 1978;100(9):2725-30. doi:10.1021/ja00477a026
Brynestad J, Smith GP. Evidence for a three-coordinate complex of nickel(II). J Am Chem Soc. 1970;92(10):3198-9. doi:10.1021/ja00713a054
Oye HA, Gruen DM. Cobalt(II) species in fused chloride solvents. Inorg Chem. 1965;4(8):1173-80. doi:10.1021/ic50030a019
Sandler RA, Larionov AA, Kostyukov AA, Troitskii AL. Behavior of iron in chloroaluminate melts in the presence of metallic aluminum. Russ J Appl Chem. 1984;10:2375.
Posypaiko VI, Alekseeva EA, editors. Diagrammy plavkosti solevykh sistem. Troynyye sistemy [Charts of salt systems' fusion. Triple systems]. Moscow: Chemistry; 1977. 328 p. Russian.
Oye HA, Gruen DM. Octahedral absorption spectra of the dipositive 3d metal ions in molten aluminum chloride. Inorg Chem. 1964;3(6):836-41. doi:10.1021/ic50016a011
DOI: https://doi.org/10.15826/chimtech.2014.1.2.699
Copyright (c) 2014 V. V. Karpov, V. A. Volkovich, I. B. Polovov, O. I. Rebrin

This work is licensed under a Creative Commons Attribution 4.0 International License.
Chimica Techno Acta, 2014-2024
ISSN 2411-1414 (Online)
Copyright Notice






